A 2025 randomized trial found that men who took the supplement Impryl for six months had no improvement in semen quality or pregnancy rates, and in some cases slightly worse outcomes.
Supplements are often marketed to men to improve sperm quality and boost pregnancy chances. The idea is that oxidative stress can damage sperm, and some supplements might protect against it.
However, results have been mixed. Smaller studies have shown small improvements in sperm quality, while larger, well-designed trials have found little to no effect on pregnancy outcomes.
The SUMMER trial, published by de Ligny et al. (2025), was designed to provide an answer by testing a widely used supplement (Impryl) in a randomized controlled trial (RCT). Nearly 1,200 men were enrolled and randomly assigned to take either Impryl or a placebo once daily for six months while trying to conceive unassisted or during fertility treatment (IUI or IVF/ICSI).
Impryl is widely marketed in the UK for preconception use, and users are advised not to take antioxidant supplements such as CoQ10, vitamin C, or selenium while using it. It’s a metabolic supplement and its ingredients include betaine 200 mg, L-cystine 200 mg, niacin 16 mg, zinc 10 mg, vitamins B6 1.4 mg & B2 1.4 mg, folate 400 µg, B12 2.5 µg.
Note: After publication, several researchers and industry representatives posted comments on the JAMA Network Open website questioning Impryl’s classification and whether the study population or treatment timing may have influenced the results. The authors responded and noted that future trials should focus on men with confirmed oxidative stress, as supplements may only benefit specific subgroups. The journal has not issued any corrections or retractions. In light of these comments, I’ve updated this post to avoid implying Impryl as an antioxidant supplement. The summary below reflects the findings as published.
🔗 Original studies are referenced in this post or within the linked Remembryo posts.
💡 Reminder: Terms underlined with a dotted black line are linked to glossary entries. Clicking these does not count toward your paywall limit.
Study details
- Study type: Multicenter, double-blind, randomized controlled trial that took place at 21 hospitals and fertility clinics in the Netherlands between 2018–2024
- Participants: Men seeking fertility care after 12 months of trying, who continued trying to conceive unassisted (about 27%), had IUI (32%) or IVF/ICSI (37%).
- There were 1171 men (591 supplement, 580 placebo); median age 34; female partners 32 years.
- Intention-to-treat population: 1171 participants (all men included)
- Per-protocol population: 680 participants (only those who followed the protocol and took ≥75% of tablets, and did not use antioxidants)
- There were 1171 men (591 supplement, 580 placebo); median age 34; female partners 32 years.
- Eligibility: No testicular sperm extraction, donor gametes, endocrine/urologic causes, or antioxidant use within 3 months
- Intervention: Daily placebo tablet or single Impryl tablet for 6 months.
- Primary outcome: Ongoing pregnancy within 6 months
- Data reporting: Patients self-reported pregnancy outcomes and how much medication they had left online in a monthly questionnaire.
- Statistical adjustment: All the adjusted odds ratios were adjusted for randomization strata (type of fertility treatment) and female age.
- Potential conflict of interest: This study was financially supported by Goodlife Pharma BV, which developed Impryl and sells it.
Pregnancy rates were similar with or without Impryl
In this study, all participants took Impryl or a placebo daily for six months, during which conception could occur either unassisted or through IUI or IVF/ICSI. The study measured how many ongoing pregnancies occurred within this six-month treatment period and continued to follow couples for another three months to capture any additional pregnancies that developed soon after.
After 6 months of taking Impryl or placebo, ongoing pregnancy rates were similar between the two groups. In the Impryl group, 193 of 571 men (33.8%) achieved an ongoing pregnancy compared with 208 of 555 (37.5%) in the placebo group. This difference was not statistically significant (adjusted odds ratio [95% CI]: 0.85 [0.66–1.09]; p= 0.20).
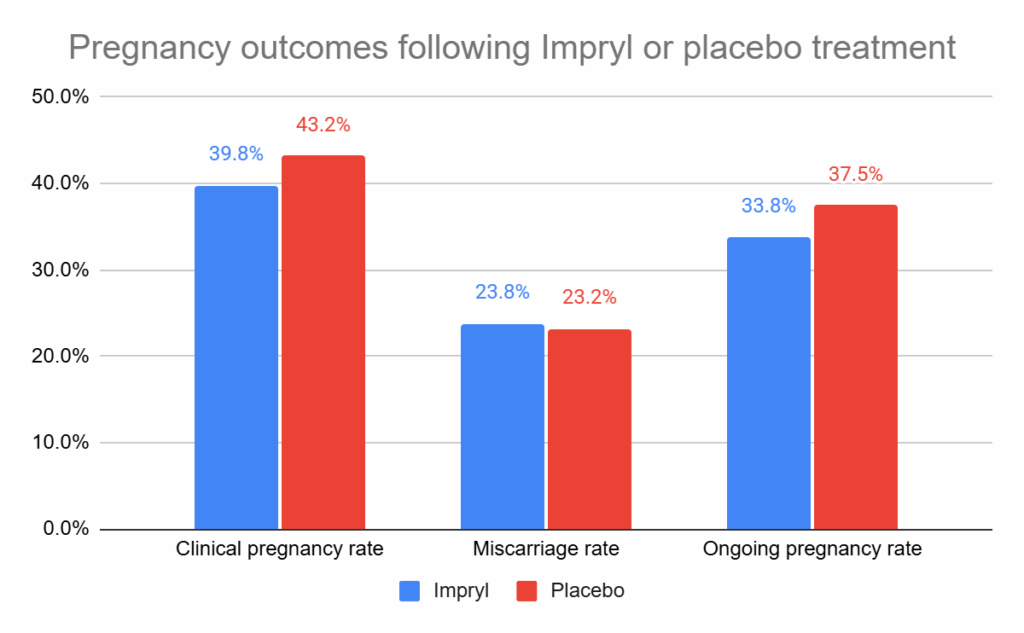
The above results are based on the intention-to-treat analysis, which included all participants who were randomized. However, adherence to treatment was relatively low — only about 58% of men took at least 75% of their tablets. When the researchers analyzed just these patients (the per-protocol group), the results were similar. This suggests that even among those who followed the regimen closely, the supplement did not improve pregnancy outcomes.
Because some men could have conceived early after only a month or two of Impryl use, the researchers also examined those who conceived between four and six months, when sperm would have fully developed under supplement exposure (about 72 days, or 2½ months). During this “optimal treatment window,” pregnancy rates were actually lower among men taking the supplement compared with placebo (15.5% vs 21.5%; adjusted odds ratio [95% CI]: 0.66 [0.47–0.94]; p= 0.02).
Overall, the supplement did not improve pregnancy outcomes, and in the subgroup where sperm had fully developed under treatment, results were slightly worse than with placebo.
Impryl did not improve IVF outcomes
This study included couples that were pursuing different fertility treatments, ranging from unassisted conception to IUI, IVF, or ICSI. The researchers analyzed results specifically for those who underwent IVF or ICSI, with 177 men in the supplement group and 169 in the placebo group.
- Fertilization rates were similar (68% vs 64%, p= 0.23).
- Embryo utilization rates were also similar (50% vs 48%, p= 0.27). This tells us how many embryos were transferred or frozen, and shows that there’s no difference in embryo quality/development between the groups.
- Pregnancy rates were lower with Impryl (57.8% vs 68.6%, p= 0.03).
- Fresh transfer pregnancy rates were lower with Impryl (42.9% vs 56.2%, p= 0.009). About 85-90% of transfers were fresh in the IVF/ICSI group.
- Frozen transfer pregnancy rates were similar (53.2% vs 62.1%, p= 0.45).
Overall, the supplement did not improve fertilization or embryo development and was linked to lower pregnancy rates after fresh transfer. There was no mention of an optimal treatment window here, so some patients may have started IVF/ICSI within a short period of exposure to the supplement.
Impryl did not improve semen quality
- Semen parameters, including volume, concentration, motility, and total motile sperm count, were similar between groups after 3–6 months of treatment.
- In the smaller subgroup tested for sperm vitality and DNA fragmentation, no differences were found in sperm concentration, motility, or DNA fragmentation.
- Sperm vitality decreased slightly after Impryl use (median 62.7% to 54.9%; p = .03), suggesting a minor reduction in the proportion of live sperm cells, although the clinical significance of this is unclear.
Conclusions
In this trial, men who took the supplement Impryl for six months did not show any improvement in pregnancy rates, semen quality, or sperm DNA integrity compared with placebo. They actually found that pregnancy rates were slightly lower during the period when the supplement should have had its strongest effect (four to six months).
Previous meta-analyses have suggested that supplements might improve pregnancy outcomes in men, but those studies were small and of lower quality. The study presented here is higher quality and the authors note that it’s the first to observe a lower ongoing pregnancy rate.
The authors suggest that the findings might reflect “reductive stress,” a state where excess reduction capacity could disrupt normal redox balance and harm sperm function. Although participants did not exceed recommended daily doses, the study did not measure baseline oxidative stress, so it’s possible that some men with already low oxidative stress were negatively affected. Most participants had normal semen parameters, suggesting they may not have had a deficiency or imbalance that the supplement could address.
Although evidence is limited, other studies suggest that taking too many male fertility supplements could be harmful for some men. Some experts recommend measuring baseline oxidative stress levels to avoid overdosing, while others have even proposed that excessive supplementation might cause infertility in certain cases. I go deeper into this topic in my post Can too many supplements harm male fertility? So far, I’ve only seen this effect reported in men, although it’s possible that similar mechanisms could apply to female fertility as well.
This isn’t the first time these researchers have questioned the evidence behind supplements. In a 2023 study (reviewed here), the same author found that fewer than half of male fertility supplements sold online had ever been tested in a clinical trial, and most contained doses exceeding the recommended daily allowance.
They write, “Patients and clinicians are often not aware of potential adverse effects and tend to assume that if it does not help, it does not hurt. Information on the efficacy and risks of nutritional supplements is currently not evidence-based and remains unclear.”
Limitations: Adherence was moderate (about 58%), limited patients with male-factor infertility (~30% — this study was done in the general population), oxidative stress wasn’t measured, medication adherence and pregnancy data was self-reported.
Want to read more about fertility supplements?
While multiple studies have shown the benefit of antioxidant supplements in improving male fertility, what effect excessive supplementation has isn't clear. This post combines a new study with older studies that show high levels of supplements can harm male fertility. However, these studies all have their limitations, and better quality research is needed to draw conclusions. Read more.
Researchers in a 2023 study evaluated 34 common male fertility sperm supplement brands for safety and efficacy. They found that the majority exceeded the recommended daily allowance, with two that exceeded the tolerable upper intake level. Less than half had supporting studies for their use, with the overall quality of evidence being ranked as poor. Read more.
This post discusses the use of supplements to improve egg quality for IVF success, exploring the supporting evidence for a range of supplements like N-acetyl-cysteine, Melatonin, L-arginine, Myo-inositol, CoQ10, L-carnitine, DHEA, folic acid/folate, and vitamins E, C, D, and B. Read more.
Researchers in a 2020 study report on the results of the MOXI trial that tested the effectiveness of a sperm supplement cocktail (containing vitamin C, vitamin D, vitamin E, selenium, and more) and found no benefit. Read more.
Related studies
These additional studies were referenced by the authors of the paper and haven’t been covered on Remembryo. They may be helpful if you’re exploring this topic further. This section is available for paid subscribers.
Reference
If you liked this post and want to support what I do, please consider a paid subscription, Patreon or donate through PayPal!

About Embryoman
Embryoman (Sean Lauber) is a former embryologist and the founder of Remembryo, an IVF research and fertility education website. After working in an IVF lab in the US, he returned to Canada and now focuses on making fertility research more accessible. He holds a Master’s in Immunology and launched Remembryo in 2018 to help patients and professionals make sense of IVF research. Sean shares weekly study updates on Facebook, Instagram, and Reddit regularly. He also answers questions on Reddit or in his private Facebook group.














