A new study used a mathematical model to simulate embryo biopsies, finding that fully euploid embryos are rare, while aneuploid or mosaic embryos were more common.
PGT-A is commonly used in IVF to select embryos with the best chance of success. One of the problems with PGT-A is that it uses a single biopsy of about 5-10 cells to represent the whole embryo, which could be hundreds of cells in size.
Embryos with both euploid and aneuploid cells are called mosaic. However, determining the true incidence of mosaicism is difficult, because the small number of cells biopsied may not be representative of the whole embryo.
Previous research has explored the prevalence of mosaicism by isolating and sequencing cells from individual embryos. These studies consistently found high rates of mosaicism: one study reported that 82% of blastocysts displayed mosaicism, while another found it in 100% of blastocysts. You can read more about these studies in my posts Mosaicism much more common than previously thought, using more sensitive PGT-A technique and More sensitive PGT-A test shows that nearly all blastocysts, fetal tissues are mosaic.
In a new study, Yang et al. (2024) used mathematical modeling to predict how common mosaic embryos are. Note that this study is currently a preprint and published to the BioRxiv server (it has not been peer reviewed).
🔗 Original studies are referenced in this post or within the linked Remembryo posts.
💡 Reminder: Terms underlined with a dotted black line are linked to glossary entries. Clicking these does not count toward your paywall limit.
The researchers focused on two types of errors: meiotic errors, which happen during the formation of eggs or sperm and can affect all cells of the embryo, and mitotic errors, which occur after fertilization as the embryo grows and typically affect only some cells, leading to mosaicism.
Below you can see how a mosaic embryo is formed, after a mitotic error occurs in one cell of a two-cell embryo. After multiple cell divisions, the abnormal cells (in red) end up representing about half of the blastocyst. They’re distributed more or less randomly in this diagram.
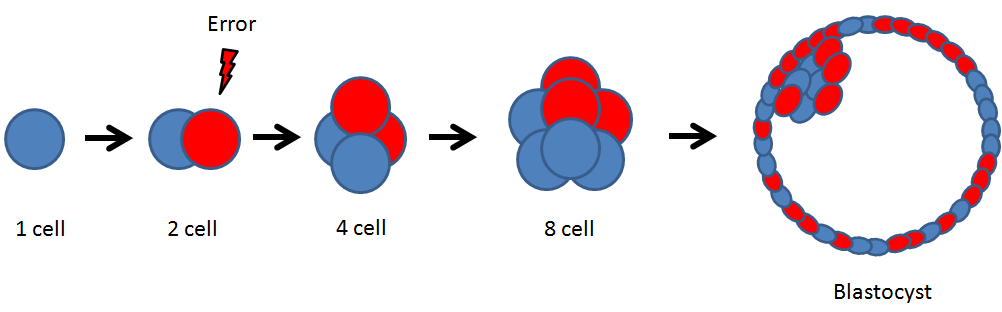
Here’s a basic outline of what the researchers did:
- The team used a software tool called Tessera to create simulations of human embryo development. They adjusted parameters like the rate of meiotic and mitotic errors and how randomly abnormal cells are distributed within the embryo.
- For each simulation, they set up scenarios with different levels of random distribution (dispersal) of abnormal cells: no dispersal (0), moderate dispersal (0.5), and complete randomness (1).
- They simulated taking a 5-cell biopsy from 1000 of these virtual embryos (each containing 256 cells), imitating what happens during PGT-A.
- To ensure their simulations were accurate, the researchers calibrated their models to match real-world data from the Viotti et al. (2021) study, which detailed PGT-A results for over 73,000 embryos—38.8% euploid, 18.6% mosaic, and 42.6% aneuploid. They selectively used simulations that mirrored this distribution of euploid, mosaic, and aneuploid embryos.
To be clear, they used software to simulate different rates of genetic errors (meiotic or mitotic) and how the abnormal cells were distributed in the embryos. Then they performed digital “biopsies” on these embryo simulations, and only used the simulations that closely matched what you would expect from real-world data (38.8% euploid, 18.6% mosaic, and 42.6% aneuploid).
These simulations were generated using a single digital biopsy, but they wanted to see what happens when they assessed every cell in the simulated embryos.
They discovered that regardless of how randomly cells were distributed, less than 1% of the embryos were entirely euploid (0%, 0.1% and 0.3% for dispersals 0, 0.5 and 1). In all cases, they found that about 60% of embryos were mosaic, and 40% were fully aneuploid. Although they didn’t report on the %, they noted that many mosaics were low level (having <25% abnormal cells).
So they found that fully euploid embryos were rare, as opposed to fully aneuploid embryos and mosaics. Let’s walk through this and explain what this means, using the diagram below for reference.
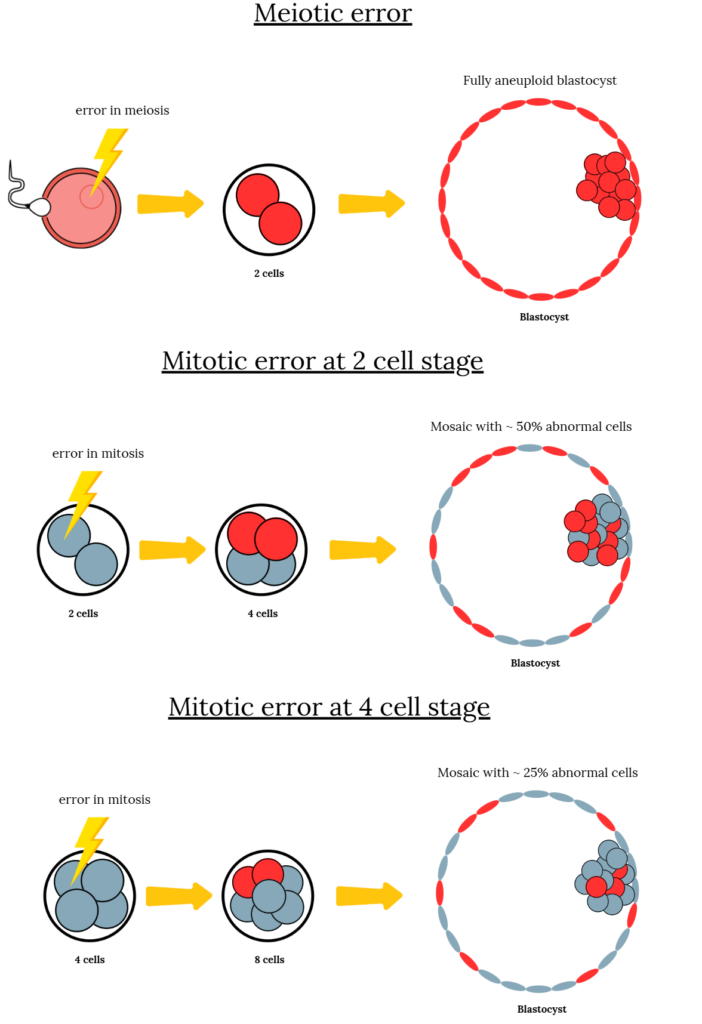
If an embryo has a meiotic error, it means there was a problem in the sperm or egg, causing all cells in the embryo to be affected and making it fully aneuploid. This leads to a high percentage of fully aneuploid embryos.
If the sperm and egg are error-free, any errors that occur after fertilization are mitotic. These can happen at any stage, such as when the embryo is at the 2-cell, 4-cell stage, blastocyst stage, etc., and can occur at a specific frequency. All cells that arise from the one with a mitotic error will be abnormal, contributing to a certain percentage of the embryo’s cells being abnormal, which determines its level of mosaicism. The further along in development the embryo is, the fewer the number of affected cells and the lower the % mosaicism.
Since mitotic errors can occur at any stage, it’s almost inevitable that every embryo will experience at least one such error, which explains why fully euploid embryos are very rare.
Conclusions
In conclusion, the researchers used a mathematical model to simulate embryo biopsies, finding that fully euploid embryos are rare while fully aneuploid and mosaic embryos were more common. This suggests that most “euploids” after PGT-A are probably low level mosaic embryos (both of which have similar success rates), and may help explain why euploids don’t lead to live births more consistently. Of course, there’s a variety of other reasons that embryos fail to implant or miscarry…
What about fully aneuploid embryos? Are they more likely to be high level mosaics?
This study shows that many embryos are fully aneuploid due to errors in the sperm or egg, though it may be possible that some of the embryo’s cells may correct after fertilization. It’s not clear how common full aneuploidy is, as far as I know. What we need is a study that sequences the DNA from every cell of aneuploid embryos. Since most clinics discard aneuploid embryos, it should be a relatively easy study to conduct. Get on it scientists!
The authors pointed out some of the limitations of this research. The study used simplified models that ignore specific chromosomes affected or self-correction. It also assumed a constant rate of cell division errors, which might not reflect how errors actually occur at different stages. There was also no consideration for embryo arrest, which may prevent an embryo from reaching the blastocyst stage. Additionally, this study has not yet been peer-reviewed, meaning it hasn’t been evaluated by other experts for accuracy and reliability.
For further reading and real-world evidence that isn’t based on a model, you can check my posts Mosaicism much more common than previously thought, using more sensitive PGT-A technique, More sensitive PGT-A test shows that nearly all blastocysts, fetal tissues are mosaic and Does a PGT-A biopsy match the rest of the embryo?
Reference
If you liked this post and want to support what I do, please consider a paid subscription, Patreon or donate through PayPal!

About Embryoman
Embryoman (Sean Lauber) is a former embryologist and the founder of Remembryo, an IVF research and fertility education website. After working in an IVF lab in the US, he returned to Canada and now focuses on making fertility research more accessible. He holds a Master’s in Immunology and launched Remembryo in 2018 to help patients and professionals make sense of IVF research. Sean shares weekly study updates on Facebook, Instagram, and Reddit regularly. He also answers questions on Reddit or in his private Facebook group.









