Researchers in a 2025 study showed that women with endometriosis or adenomyosis had lower IVF success overall after 3 IVF cycles, with more differences in the first, fresh transfer.
Endometriosis and adenomyosis are two conditions that can make fertility treatment more challenging. Both are linked to inflammation, hormone resistance, and changes in how the uterus functions.
Studies have shown that these factors may reduce the chances of implantation and pregnancy, and many fertility specialists consider them important to assess before IVF.
A study by Alson et al. (2025) examined how endometriosis and adenomyosis affect the overall chance of live birth after 3 rounds of IVF, using standardized ultrasound criteria for diagnosis (IDEA/MUSA).
🔗 Original studies are referenced in this post or within the linked Remembryo posts.
💡 Reminder: Terms underlined with a dotted black line are linked to glossary entries. Clicking these does not count toward your paywall limit.
Study details
- Study type: Prospective cohort study that took place between 2019–2024 at a hospital-based IVF center in Sweden.
- Eligibility: Ages 25–39, BMI 18–30, non-smokers, first IVF/ICSI cycle, at least one year of infertility, no children with current partner, no history of surgical treatment for endometriosis, no egg donation.
- Diagnosis: All women had a 2D and 3D transvaginal ultrasound by one expert. Diagnostic definitions were based on the International Deep Endometriosis Analysis (IDEA) and the Morphological Uterus Sonographic Assessment (MUSA) groups. Women were diagnosed as having endometrioma, deep endometriosis and/or adenomyosis.
-
Participants: There were 1,035 women in this study (average age was 32.0):
- Three cycles of IVF/ICSI were offered: 1,035 completed the first cycle (742 without endometriosis and/or adenomyosis and 293 with), 473 moved on to a second cycle (319 without and 154 with), and 217 completed a third cycle (133 without and 84 with).
- Of the women included, 742 had no endometriosis or adenomyosis, 191 had endometriosis only, 61 had adenomyosis only, and 41 had both conditions.
- Treatment: Standard IVF/ICSI with single-embryo transfer on day 2-3 or day 5-6. Surplus embryos were frozen on days 5–6. Embryos from each cycle were used until a live birth or none were left.
- Primary outcome: Cumulative live birth rate after up to 3 cycles (including all fresh and frozen transfers).
Lower cumulative live births with endometriosis, adenomyosis
When all 3 rounds of IVF were taken into account, women with endometriosis and/or adenomyosis had a lower chance of live birth compared to women without these conditions. About 53% of women with endometriosis and/or adenomyosis had a live birth by the end of the 3 cycles, compared to about 69% of women without (after adjusting for age, odds ratio [95% CI]: 0.80 [0.71-0.90], p< 0.001).
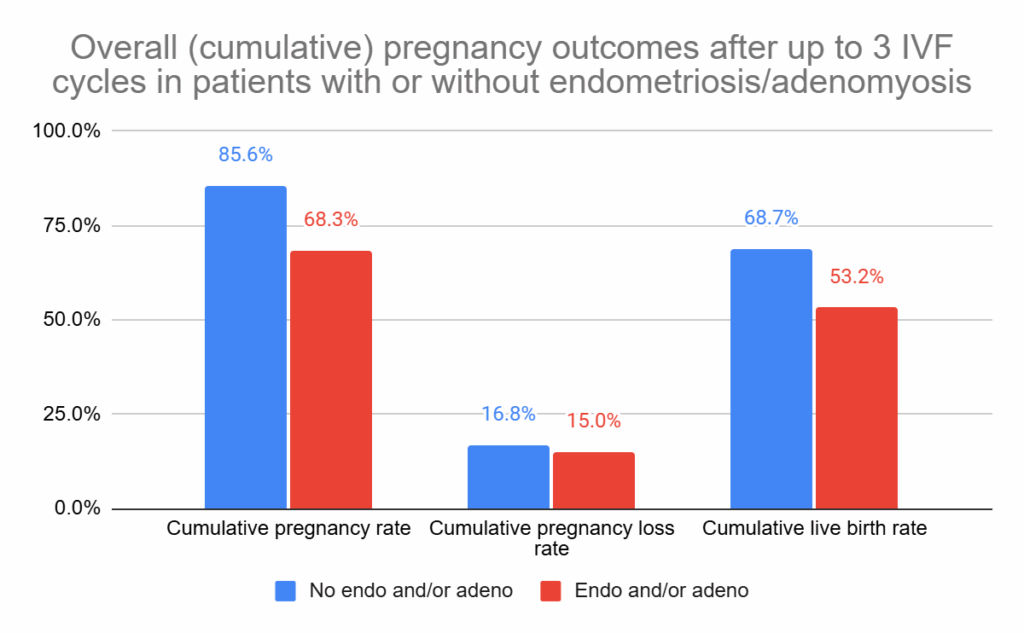
The graph above shows the intention-to-treat (ITT) results, which represent all women who started treatment regardless of whether they discontinued treatment (some dropped out). When looking only at those who completed all cycles (per-protocol), the rates were higher: 85.7% without endo/adeno and 70.0% with it (adjusted odds ratio [95% CI]: 0.85 [0.77-0.93], p< 0.001).
Bigger differences seen after fresh transfers
They also reported on the outcomes per transfer for each of the 3 IVF cycles:
- In the first cycle, 30.7% of women with endometriosis and/or adenomyosis had a live birth compared to 45.1% of those without (p< 0.001).
- In the second cycle, the numbers were 28.6% vs 41.4% (p= 0.023).
- By the third cycle, the rates were 26.2% vs 32.3%, and the difference was no longer significant (p= 0.183).
This shows that the biggest drops in live birth rates is in the early attempts, which seem to be due to larger differences in fresh vs frozen transfers:
- First cycle (fresh): 19.8% with endo and/or adeno vs 32.0% without (p< 0.001).
- First cycle (frozen): 38.0% vs 46.0% (not significant).
- Second cycle (fresh): 22.0% vs 41.4% (p< 0.001).
- Second cycle (frozen): 33.3% vs 52.9% (p= 0.042).
- Third cycle: differences were small for both fresh and frozen and not significant.
This suggests that the disadvantage was most evident in fresh cycles, possibly due to high hormone levels during stimulation, while frozen cycles appeared less affected. Another factor may be embryo stage: in the first cycle, women without endometriosis/adenomyosis had more blastocyst transfers (51% vs 40%), while those with these conditions had more cleavage-stage transfers (60% vs 49%).
Differences by endometriosis and adenomyosis subtype
In this study, some women had only endometriosis, some only adenomyosis, and some had both conditions. When comparing each group to women without disease, they found significantly lower live birth rates across all categories (p< 0.001).
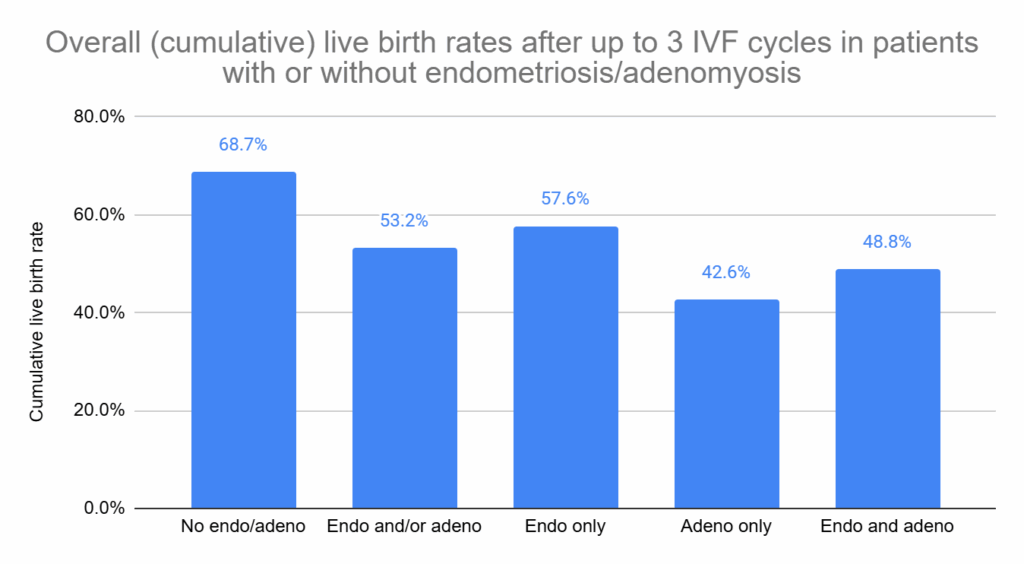
The results shown above are based on the ITT analysis. When looking at the per-protocol results (women who didn’t drop out), the rates were 85.7% for women without endo/adeno, 70.0% for those with endo and/or adeno, 73.6% for endometriosis only, 42.6% for adenomyosis only, and 48.8% for both. All comparisons remained statistically significant (p< 0.001).
For patients, this suggests that adenomyosis on its own may have the strongest impact on IVF success, while endometriosis alone has a smaller but still measurable effect.
When the researchers looked more closely at different types of endometriosis, they found that deep endometriosis on its own did not significantly affect cumulative live birth rates. In contrast, endometriomas were linked to lower success (67.6% vs 83.7%), and women who had both endometriomas and deep endometriosis had outcomes in between (71.7% vs 83.7%).
Conclusions
Women with endometriosis or adenomyosis had lower chances of live birth after IVF compared to women without these conditions. By the end of three cycles, about 53% of women with endometriosis and/or adenomyosis had a live birth versus nearly 69% without (ITT analysis). When looking only at women who completed all three cycles (per-protocol), the rates were higher — 70% with endometriosis/adenomyosis and 86% without.
The difference was strongest in the first cycle, especially after fresh transfers, where outcomes were much lower, while frozen transfers showed smaller gaps. By the third cycle, the differences were no longer significant. The authors note that women with endometriosis or adenomyosis were more often switched to the agonist protocol in later cycles, which has been linked to higher pregnancy rates in endometriosis. This change likely reflects what happens in real life, where outcomes improve as treatment protocols are adjusted across cycles.
When looking at subtypes, outcomes were reduced for endometriosis, adenomyosis, and both together, with adenomyosis showing the strongest impact.
Previous studies have linked endometriosis and adenomyosis to lower IVF success, often due to inflammation, impaired implantation, or reduced endometrial receptivity. However, some studies have found no association, which the authors suggest may be explained by differences in study design or diagnostic methods. Another issue is that studies using laparoscopy for diagnosis (the gold standard) may also include women who had surgical treatment at the same time, making it harder to separate the effect of the condition itself from the impact of treatment.
Diagnosis was made with standardized ultrasound (IDEA/MUSA) by an experienced examiner, which is considered reliable but still depends on the operator. Laparoscopy can provide more detailed information, but it involves surgery and is not usually part of the standard infertility workup.
Limitations: No surgical confirmation, possible under-detection of mild disease, PGT-A not used, cleavage stage and blastocyst transfers allowed, and findings may be less generalizable outside specialized ultrasound centers.
Want to read more about endometriosis/adenomyosis and IVF?
A 2025 study found that PCX, a surface molecule on endometrial cells, may lead to a shorter implantation window in women with endometriosis and might explain the stickiness of lesions outside the uterus. Read more.
A 2024 study combined studies involving unexplained infertility patients and endometriosis, finding that nearly half of them have the condition. Read more.
Researchers in a 2024 study found lower cumulative live birth rates in patients with a particular type of endometriosis (DIE and/or ovarian endometrioma) that was diagnosed using transvaginal ultrasound. Read more.
Researchers in a 2022 study found that the combination of endometriosis and adenomyosis results in lower cumulative live birth rates after IVF, particularly in those with T2 high-signal intensity myometrial spots as evaluated by MRI. Read more.
Related studies
These additional studies were referenced by the authors of the paper and haven’t been covered on Remembryo. They may be helpful if you’re exploring this topic further. This section is available for paid subscribers.
Reference
If you liked this post and want to support what I do, please consider a paid subscription, Patreon or donate through PayPal!

About Embryoman
Embryoman (Sean Lauber) is a former embryologist and the founder of Remembryo, an IVF research and fertility education website. After working in an IVF lab in the US, he returned to Canada and now focuses on making fertility research more accessible. He holds a Master’s in Immunology and launched Remembryo in 2018 to help patients and professionals make sense of IVF research. Sean shares weekly study updates on Facebook, Instagram, and Reddit regularly. He also answers questions on Reddit or in his private Facebook group.