A 2025 study found that RIF and RPL patients with abnormal EMMA & ALICE results that had antibiotic and/or probiotic treatment showed similar pregnancy rates to those with normal EMMA & ALICE results.
Patients with recurrent implantation failure (RIF) have consistent implantation failure due some underlying factor.
The vaginal and endometrial microbiome can affect reproductive health and some data has shown that probiotics containing Lactobacillus can improve fertility outcomes. Additionally, the presence of certain bacteria can lead to chronic endometritis that’s associated with increased risk of pregnancy loss.
In order to evaluate the endometrial microbiome, two tests can be done called EMMA & ALICE. These tests analyze the microbial composition of the endometrium, allowing patients to take steps to improve its health, such as using antibiotics to eliminate harmful bacteria or probiotics to boost beneficial Lactobacillus populations.
This post is a summary of a study by Iwami et al. (2025), who evaluated the use of EMMA & ALICE in RIF and recurrent pregnancy loss (RPL) patients. Patients who had abnormal EMMA & ALICE results received antibiotic and/or probiotic treatments before embryo transfer.
For more background information, check my post The impact of the vaginal and endometrial microbiome on reproductive health and IVF outcomes.
🔗 Original studies are referenced in this post or within the linked Remembryo posts.
💡 Reminder: Terms underlined with a dotted black line are linked to glossary entries. Clicking these does not count toward your paywall limit.
Study details
- This was a prospective study, conducted at 14 Japanese fertility clinics between 2019 and 2021.
- Patient had more than 3 transfers with no implantation (RIF) or more than 2 miscarriages (RPL).
- Igenomix’s EMMA & ALICE procedure was followed, with an endometrial biopsy taken during the luteal phase.
- Probiotics involved vaginal suppositories given 7-10 days before embryo transfer (Invag® or Lactoflora®).
- The primary outcome was the ongoing pregnancy rate.
In terms of sample size, there were 527 patients, split into 3 groups based on their EMMA & ALICE results:
- Normal (229 patients): These patients had a Lactobacillus-dominated endometrial microbiota (>90% Lactobacillus). These patients didn’t receive any antibiotic or probiotic treatments.
- Abnormal (110 patients, with 83 ALICE negative and 27 ALICE positive): These patients had an endometrial microbiota that was not Lactobacillus-dominated (<90% Lactobacillus) and had non-Lactobacillus genera constituting 10% or more, including pathogenic bacteria linked to chronic endometritis (ALICE positive). They received targeted antibiotic therapy to eliminate the specific pathogens, followed by a confirmatory endometrial biopsy and EMMA & ALICE retesting (77 of patients required a single course of targeted antibiotics, 33 had a second course and 12 had a third course). After pathogen clearance, patients were given Lactobacillus probiotics 7-10 days before proceeding with embryo transfer.
- Mild dysbiosis + ultralow biomass (188 patients, with 114 having mild dysbiosis and 74 having ultralow biomass): These patients had an endometrial microbiota that was not Lactobacillus-dominated (<90% Lactobacillus), but the other bacteria found were in very low amounts. In ultralow biomass patients, there were very few bacteria and the endometrium was “almost sterile.” The mild+ultralow group received a Lactobacillus probiotic 7-10 days before embryo transfer.
In terms of patient characteristics, there were no differences in age (about 37-38 on average), duration on infertility, history of births, average number of transfer cycles (3), transfer of cleavage stage or blastocyst stage embryo, or use of PGT-A.
About half of RIF patients had abnormal EMMA & ALICE results
As indicated above, 527 patients received EMMA & ALICE, with 229 having a normal result, 110 with an abnormal EMMA result (83 ALICE negative and 27 ALICE positive), and 188 having either mild dysbiosis (114) or ultralow biomass (74).

In the EMMA abnormal group, the most common non-Lactobacillus genera detected was Gardnerella (41.2% of patients), followed by Streptococcus (33.8%) and Atopobium (28.0%).
After treatment, abnormal EMMA & ALICE patients achieved similar pregnancy outcomes to normal patients
Patients with abnormal EMMA results, or who had mild dysbiosis or ultralow biomass, were treated as indicated in the Study Details section before their embryo transfer. All the groups had similar pregnancy outcomes, with no statistical differences.

Patients with abnormal EMMA & ALICE results who received antibiotics and/or probiotics achieved similar pregnancy outcomes to those with normal results, suggesting that the treatment improved outcomes for these abnormal patients—who would likely have had lower pregnancy rates without intervention.
But this is kind of cheating, since we don’t know for certain that these abnormal EMMA patients would have lower pregnancy rates. What’s missing here is a control group that didn’t receive EMMA & ALICE testing, or even a group of abnormal EMMA patients who received no probiotic treatment (although this wouldn’t be ethical). This is a big limitation of the study.
However, the same research group performed a pilot study that included a control group, so let’s look at that below.
No control group? A look at a previous study by the same group
The same authors did publish a smaller pilot study in 2023 that included a control group. The protocol was essentially the same as the larger study presented here, but only included EMMA and no ALICE (they used hysteroscopy to detect chronic endometritis). Here’s a breakdown of the groups in this study:
- No EMMA (67 patients)
- EMMA (131 patients): EMMA normal (67), EMMA abnormal (30), mild dysbiosis or ultralow biomass (44). The ratios here are about the same as the larger study, which is nice!
Pregnancy outcomes for the two groups (EMMA vs no EMMA) were compared. The group that had EMMA and antibiotic and/or probiotic treatment showed improved ongoing pregnancy rates vs no EMMA (33.6% vs 9.4%, p< 0.001).
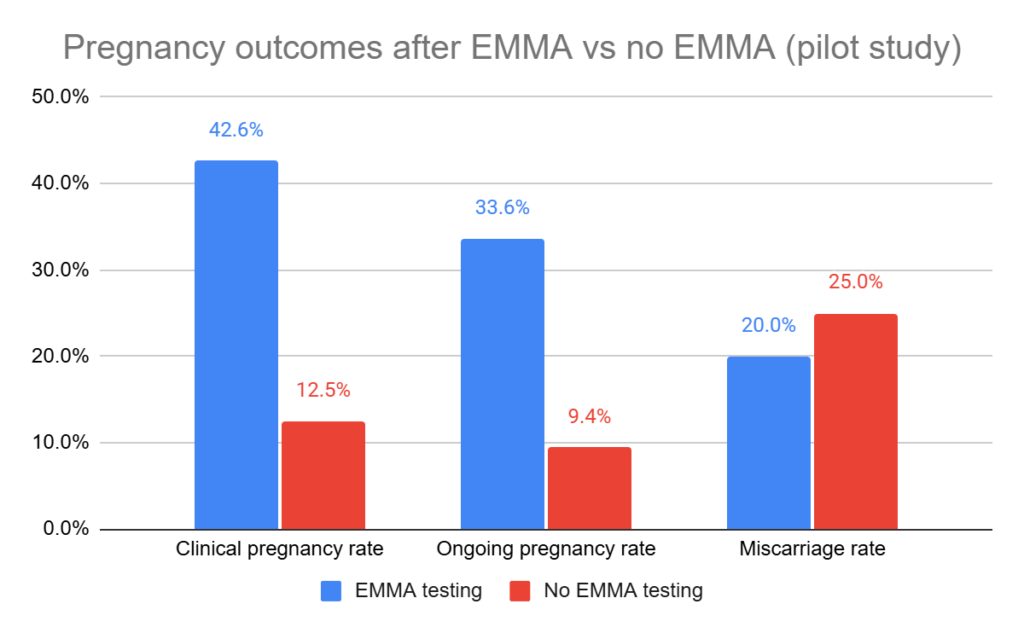
The ongoing pregnancy rate for the no EMMA group is very low, which surprises me, but the smaller sample size could be playing a role. They also looked at cumulative ongoing pregnancy rates after 2 transfers for EMMA vs no EMMA, with the EMMA group still having a higher rate (48.9% vs 32.8%, p= 0.028).
Having a pilot study that includes a control group is nice to see, but it doesn’t make up for them not including it in the larger study!
Older patients may benefit more from antibiotic, probiotic treatment
Back to the original, larger study now!
The researchers also evaluated the difference in cumulative ongoing pregnancy rate after up to 12 months (although it wasn’t clear how many IVF cycles each group had, the number of transfers, or how many patients dropped out). Here are the cumulative rates after 12 months, which I approximated from a graph:
- EMMA normal: about 70%
- EMMA abnormal (ALICE negative): about 70%. This group achieved pregnancy earlier than the other groups (p= 0.02).
- EMMA abnormal (ALICE positive): about 40%
- Mild dysbiosis: about 60%
- Ultralow biomass: about 60%
They also looked at different age groups and measured the cumulative ongoing pregnancy rates:
- Less than 35: no differences (all about 60% after 9 months).
- 36-40: those with abnormal EMMA results had a higher cumulative ongoing pregnancy rate (about 75% vs about 60% for normal EMMA after 12 months, p= 0.036; the mild dysbiosis/ultra group was about 80%).
- Over 41: those with abnormal EMMA results had a higher cumulative ongoing pregnancy rate (about 55% vs about 35% for normal EMMA after 12 months, p= 0.03; the mild dysbiosis/ultra group was about 45%).
This shows that older patients (over 35) with abnormal EMMA results have a higher chance of pregnancy after antibiotic and/or probiotic treatment compared to those with normal EMMA results (who didn’t receive antibiotic and/or probiotic treatment). This suggests that antibiotic and/or probiotic treatment might correct imbalances that even EMMA normal patients have, particularly in older women.
Conclusions
The researchers found that about half of RIF and RPL patients had abnormal EMMA & ALICE results. These patients should hypothetically have lower pregnancy outcomes because of their non-ideal microbiome.
These patients received antibiotic and/or probiotic treatment, and they had similar results as patients with normal EMMA & ALICE results who didn’t have antibiotic and/or probiotic treatment. This suggests that the treatment improved outcomes for abnormal EMMA & ALICE patients, although there was no control group so it’s hard to be certain.
They also found that older patients with abnormal EMMA and antibiotic and/or probiotic treatment had higher cumulative ongoing pregnancy rates, suggesting that this treatment might correct other imbalances found in older women.
Besides the lack of the control, there were some other issues with this study…
Igenomix, the manufacturer of the EMMA & ALICE test, was listed as a funder of the 2025 research. This means they likely provided the test free of charge for the patients, but they may have also played a role in data analysis or interpretation. The lack of clarity on Igenomix’s specific role means we need to be cautious when interpreting the results.
The 2023 pilot study was registered in March 2019 while the 2025 multicenter study was registered in June 2019. This suggests that soon after the pilot was initiated, potentially due to positive preliminary results, the larger study was registered with funding from Igenomix. There was also overlap between the studies, with the pilot study including patients from March 2019 to April 2021, and the 2025 multicenter study including patients from July 2019 to August 2021. Since both studies included the same center, this overlap raises questions about how patients were selected and whether some of the same patients were used in both studies, potentially inflating the effectiveness of the treatment. I don’t think this happened, but transparency here would have been nice!
The authors even published a third study, which seems like an earlier version of the pilot study. I can’t find the full article, but there’s a version of it on European Medical Journal website. In this study they recruited 158 patients between July 2018 and March 2020 at their center. In the study, they found an improved pregnancy rate for patients with EMMA & ALICE vs those without it (64.5% vs 33.3%).
Has there been any other research on the topic?
The only other study I could find was an abstract-only study from Bamford et al. (2022), who tested EMMA & ALICE in about 100 patients at 22 clinics in the UK. EMMA resulted in a normal result in 36% of patients, 22% were abnormal, 17% had ultra-low biomass and 25% mild dysbiosis. They didn’t find any difference in pregnancy rates for patients with abnormal EMMA results.
Clearly, more data is needed!
For more information, check these posts:
- The impact of the vaginal and endometrial microbiome on reproductive health and IVF outcomes
- Patients with antibiotic-cured endometritis still show increased pregnancy loss
- RCT finds probiotic supplementation in women without RIF may be beneficial
Reference
If you liked this post and want to support what I do, please consider a paid subscription, Patreon or donate through PayPal!

About Embryoman
Embryoman (Sean Lauber) is a former embryologist and the founder of Remembryo, an IVF research and fertility education website. After working in an IVF lab in the US, he returned to Canada and now focuses on making fertility research more accessible. He holds a Master’s in Immunology and launched Remembryo in 2018 to help patients and professionals make sense of IVF research. Sean shares weekly study updates on Facebook, Instagram, and Reddit regularly. He also answers questions on Reddit or in his private Facebook group.