A 2024 study compared outcomes for patients with or without PGT-A, finding that cumulative live birth rates increased mostly for patients aged 38 and older.
PGT-A is a technique that involves testing the DNA of embryos to help select an embryo for transfer. Euploid embryos, with the right number/structure of chromosomes, are thought to have higher success rates and a lower chance of miscarriage.
This post is a summary of a study by Harris et al. (2024), who compared cumulative live birth rates (CLBR) and other outcomes for patients with or without PGT-A having their first IVF cycle. These patients were good prognosis, 40 years of age or younger, and had more than 5 fertilized eggs.
For more background on PGT-A, check my Complete guide to PGT-A.
🔗 Original studies are referenced in this post or within the linked Remembryo posts.
💡 Reminder: Terms underlined with a dotted black line are linked to glossary entries. Clicking these does not count toward your paywall limit.
Study details
This section covers key details of how the study was performed, including patient characteristics, how they were treated, and other methods used. For those who aren’t interested in these details, and just want to see the results, you can go ahead and skip this part.
- This was a retrospective study that used SART data that included most US clinics between 2016 and 2019.
- Patients were 21-40 years old, having their first IVF cycle with or without PGT-A (with NGS). These were good prognosis patients.
- Exclusions: No donor gametes, no recurrent pregnancy loss, no patients with <5 fertilized eggs.
- Primary outcome was CLBR, after up to 3 single blastocyst transfers from the patients’ first IVF cycle. Patients doing PGT-A that didn’t get any euploids, and therefore couldn’t transfer embryos, were included in the analysis as having no cumulative live birth.
In terms of sample size, there were 56,469 first IVF cycles included, with 12.2% being PGT-A. Here’s a breakdown of the age groups and number of patients with or without PGT-A:
- Aged <35: 39,802 (38,155 without PGT-A, 1647 with PGT-A).
- 35-37: 11,555 (9152 without PGT-A, 2403 with PGT-A).
- 38-40: 5112 (2301 without PGT-A, 2811 with PGT-A).
Here’s a look at how many eggs were retrieved and blastocysts from the patients’ first cycle:
- <35: 19.4 eggs retrieved and 6.3 blastocysts.
- 35-37: 17 eggs retrieved and 5.4 blastocysts.
- 38-40: 15.7 eggs retrieved and 4.5 blastocysts.
In general, patients with PGT-A had slightly more eggs retrieved and more blastocysts compared to patients without PGT-A.
Patients with PGT-A are more likely to have no embryos transferred
This study included patients’ who had their first IVF cycle between 2016-2019, with or without PGT-A. Only patients with 5 or more eggs retrieved were included, and only the first 3 transfers were considered.
While all the patients without PGT-A had an embryo transfer, some patients with PGT-A had no embryos to transfer. This increased with age from about 3% for patients aged <35 to nearly 20% for those aged 38-40. Note that a limitation of this study is that they couldn’t confirm that these patients in fact didn’t have euploids to transfer, although this is the most likely scenario. More details in the conclusion.
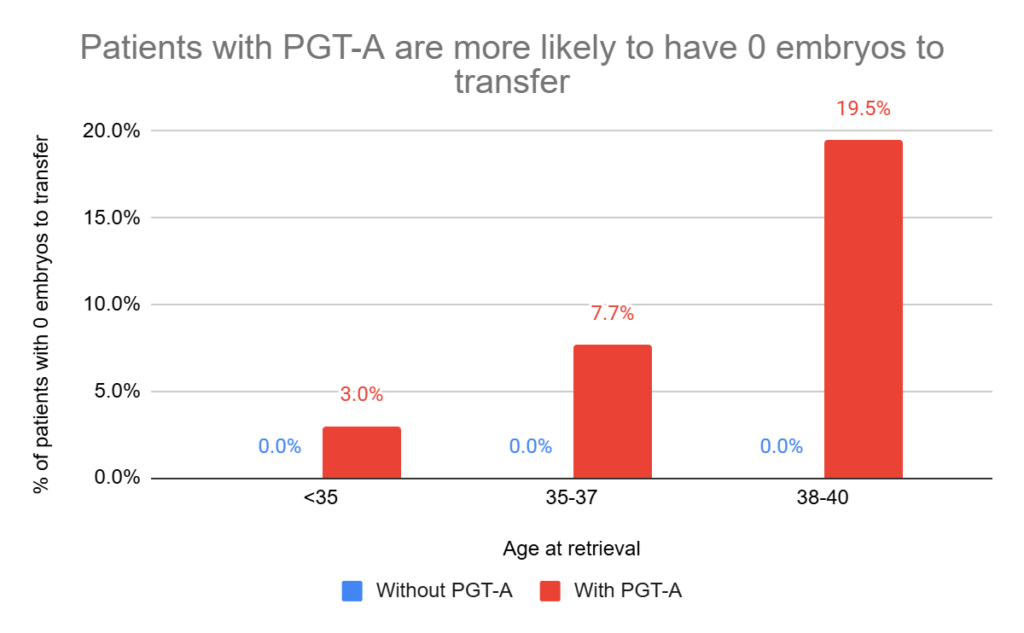
This makes sense, since the patients with PGT-A are only able to transfer embryos if they’re euploid, and the chance of a euploid embryo decreases with age (discussed in my post A look at how PGT-A results change with age, using data from over 86,000 biopsies). Why does aneuploidy increase with age? This is mainly due to errors in egg development, as discussed in my post Chromosomal errors in IVF: What is aneuploidy and what causes it?
How many patients had 1, 2 or 3 single blastocyst transfers? I’ve listed all these % in the chart below, which shows the chances of needing a second or third transfer if the previous one didn’t result in a live birth. Remember, they only looked at a max of 3 transfers of a single blastocyst from a patient’s first IVF cycle, and all these cycles had 5 or more fertilized eggs!!
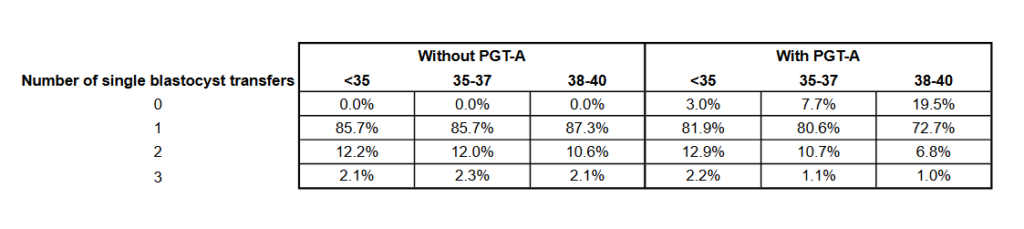
For patients without PGT-A, the number of transfers was similar between ages, while for patients with PGT-A, the number of transfers changed more with age. This is probably because they’ve run out of euploids to transfer after a first or second transfer.
Cumulative live birth rate increases with PGT-A, but only for older patients
Now let’s look at the CLBR! This was the primary outcome, or the “main point” of this study. It’s the overall % of patients who had a live birth, whether it took a single transfer or up to 3. It’s including all the patients, whether or not they had euploids to transfer.
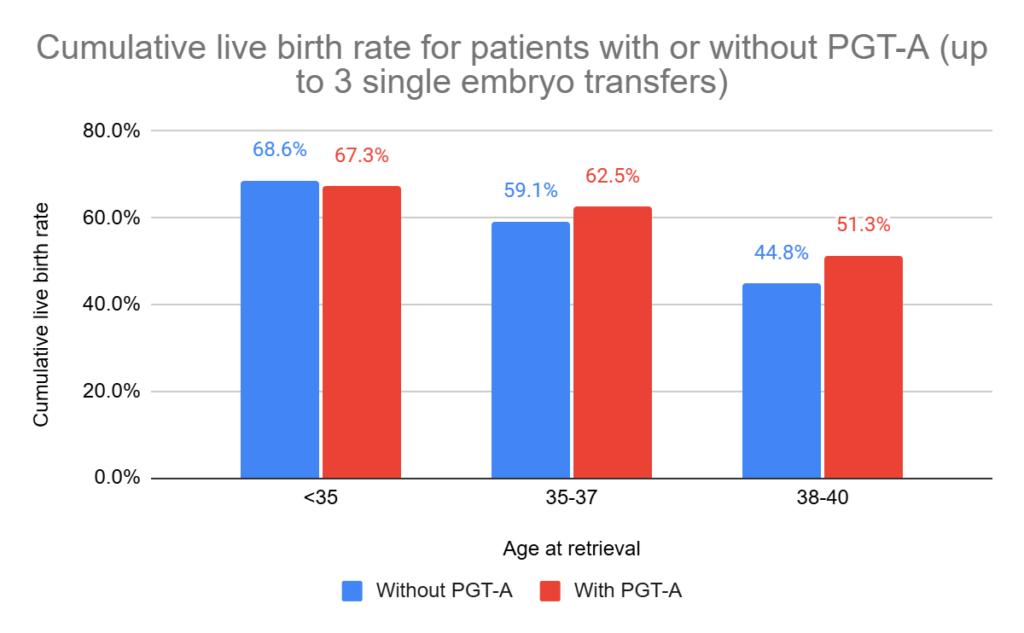
They statistically adjusted these results to account for differences in race, BMI, parity, smoking, infertility diagnosis and the number of blastocysts:
- Patients aged <35 had a slightly lower CLBR with PGT than without it (67.3% vs 68.6%, relative risk [95% CI]: 0.96 [0.93-0.99]).
- Patients aged 35-37 had a slightly higher CLBR with PGT than without it (59.1% vs 62.5%, relative risk [95% CI]: 1.04 [1.00-1.08]).
- Patients aged 38-40 had a higher CLBR with PGT than without it (44.8% vs 51.3%, relative risk [95% CI]: 1.14 [1.07-1.20]).
This shows that for patients 37 or younger, there are small differences with or without PGT-A which may not be clinically significant. This means that transferring a patient’s best quality untested embryos or euploids has about the same chance of working for patients <37. Better quality embryos tend to have a higher chance of being euploid anyway.
Patients 38-40 saw an increase in CLBR when doing PGT-A. This means that transferring euploids had a better chance of working than untested embryos for patients 38-40. It’s interesting that there was a lower CLBR here than the younger ages — this is likely because these patients have fewer euploids available for transfer.
Other pregnancy outcomes for patients with or without PGT-A
The CLBR we saw above included all patients, whether or not they had a transfer. So how do things look when we’re only considering the patients that had a euploid to transfer? The live birth rate here will obviously be higher — since it’s excluding patients that didn’t have euploids to transfer. Let’s take a look, while also considering other pregnancy outcomes like miscarriage rates!
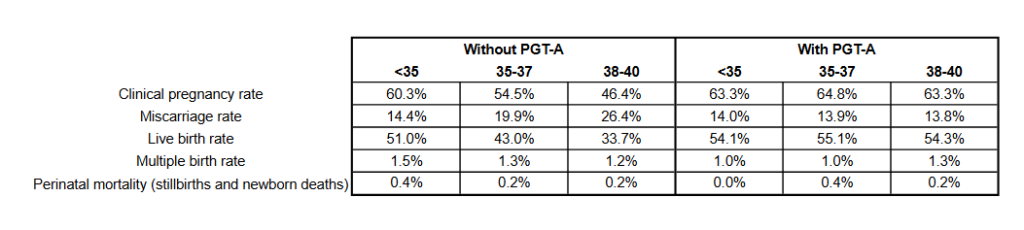
After statistical adjustment:
- Clinical pregnancies increased for patients aged 35-40 with PGT-A (not <35). For 35-37: relative risk (95% CI) = 1.16 (1.12–1.19); for 38-40: relative risk (95% CI) = 1.32 (1.26-1.39).
- Miscarriages decreased for patients aged 35-40 with PGT-A (not <35). For 35-37: relative risk (95% CI) = 0.71 (0.62–0.80); for 38-40: relative risk (95% CI) = 0.52 (0.45–0.61).
- Live birth increased for patients aged 35-40 with PGT-A (not <35). For 35-37: relative risk (95% CI) = 1.24 (1.19–1.29); for 38-40: relative risk (95% CI) = 1.55 (1.45-1.64).
Subgroup: Freeze-all cycles show somewhat similar results
The data above includes fresh and frozen transfers for the patients without PGT-A. The researchers did a separate (subgroup) analysis on just the patients who did a freeze-all cycle and didn’t have a fresh transfer.
I’m not going to get into all the details here, as the above data is larger and I think it better represents what patients are doing, but here’s what they generally found:
- CLBR didn’t increase for <35 with PGT-A but increased for 35-40 (no decrease in CLBR for <35 like above).
- Live birth increased for all ages with PGT-A (not just for 35-40 as shown above).
- Miscarriage decreased only for those 35-40 (like the above data).
Conclusions
For patients under 35, PGT-A showed a slight decrease in CLBR compared to no PGT-A, and for ages 35–37 there was a slight increase. I feel like these minor differences aren’t all that clinically significant, and there isn’t much of a difference in transferring a euploid or a best quality untested embryo for patients <38.
However, for patients 38–40, PGT-A clearly improved the CLBR. This shows that transferring euploids leads to more live births than untested embryos for this age group.
Miscarriage rates were similar for patients <35, but decreased with PGT-A for patients aged 35-40.
This isn’t the first study to show this:
- Munne et al. (2019) used NGS for PGT-A and found no difference in ongoing pregnancy rates between patients who transferred a euploid or their best quality embryo. A post hoc analysis found that women aged 35-40 had an increased ongoing pregnancy rate with a euploid transfer (51% vs 37%). I reviewed this study in my post PGT-A doesn’t improve success rates in good prognosis patients.
- Yan et al. (2021) used NGS for PGT-A and found no difference in CLBR after up to three embryo/euploid transfers within 1 year (77.2% vs 81.8%). I reviewed this study in my post PGT-A vs embryo grading: results of a 2021 clinical trial.
However, using these results to say that PGT-A isn’t effective for patients <38 is a bit misleading, because the true benefit of PGT-A may be in identifying aneuploids that won’t be transferred, and this would be useful even for younger patients. An analysis by Capalbo et al. (2022) shows that about 2% of them lead to live births, so PGT-A seems to be a pretty effective tool in identifying embryos with a very low chance of working. However, this is based on limited data, and it’s possible that there are differences based on age. With inaccuracies in PGT, some aneuploids could be mosaic, and maybe younger patients are better equipped to handle self-correction? In other words, do younger patients have higher live birth rates with aneuploid transfers compared to older patients? We need more data on transferring aneuploid embryos to know for sure!!
Let’s look at some issues with this study!
An issue with this study is that it looked at a specific group of patients that doesn’t really represent those who PGT-A is intended for: they were younger, having their first cycle and didn’t have a history of recurrent losses. Patients opting for PGT-A may do so because they’re on their 3rd cycle, and have had multiple failed transfers.
But for patients having their first IVF cycle who are younger than 38? This study shows that PGT-A might not make sense, especially when you consider how expensive it is and some of the other problems with PGT-A.
Another issue with this study is that it only considers 3 transfers, but a true CLBR should account for all embryos from a single cycle. If PGT-A perfectly identifies all viable embryos, the CLBR would be the same; if not, it could be lower since some viable embryos might be missed. The second point here is more realistic, since PGT-A definitely does not perfectly identify non-aneuploid embryos (again, check my post on the problems with PGT-A). But we’re not going to see this kind of result unless all embryos are transferred.
Besides these issues, the authors brought up some other limitations of their study:
- Mosaic embryos weren’t considered! I’m not sure if this information is included in the SART database, but including mosaic transfers would have been a good idea (or any non-aneuploid embryo really — although we wouldn’t see this kind of data from a national database since many clinics are reluctant to transfer certain mosaics).
- For people with no euploids to transfer, it’s not clear if this is because they had no euploids or if the embryo didn’t survive the thaw, or if the embryos were transferred to another clinic, or banked, etc. This information wasn’t available in the SART database.
- They excluded patients transferring more than one embryo. The ASRM recommends up to 2 for patients without PGT-A. They did this to be consistent, and this is the reason they didn’t include older patients in this study, because they’re more likely to transfer more than a single embryo.
Reference
If you liked this post and want to support what I do, please consider a paid subscription, Patreon or donate through PayPal!

About Embryoman
Embryoman (Sean Lauber) is a former embryologist and the founder of Remembryo, an IVF research and fertility education website. After working in an IVF lab in the US, he returned to Canada and now focuses on making fertility research more accessible. He holds a Master’s in Immunology and launched Remembryo in 2018 to help patients and professionals make sense of IVF research. Sean shares weekly study updates on Facebook, Instagram, and Reddit regularly. He also answers questions on Reddit or in his private Facebook group.









