This post covers PGT-A (formerly called PGS testing), including how it works, how accurate it is, whether it can damage embryos, how to read PGT-A reports, success rates, mosaic embryos, and why even euploid embryos sometimes fail.
🔗 Original studies are referenced in this post or within the linked Remembryo posts.
💡 Reminder: Terms underlined with a dotted black line are linked to glossary entries. Clicking these does not count toward your paywall limit.
Table of Contents
Definitions
Preimplantation genetic testing (PGT) is the general term for testing the DNA of embryos.
There are several versions of PGT:
- PGT-A = PGT for aneuploidy (aka PGS or CCS)
- PGT-M = PGT for monogenic/single gene defects (aka PGD)
- PGT-SR = PGT for structural rearrangements
This post will discuss PGT-A exclusively!
What is a chromosome?
Before we talk about PGT-A, it’s important that we understand what a chromosome is!
A chromosome is a single long piece of DNA that is wrapped up tightly around specialized proteins called histones to make nucleosomes. These nucleosomes are wrapped up further to make chromatin, which is wrapped up even more to make a chromosome.
To give you an idea of how much DNA is compacted inside a single cell… If you were to measure the length of DNA inside a single human cell, it would be about 2 meters long.
That 2 meters of DNA is stuffed inside the nucleus of most cells in our body — all thanks to the amazing ability of DNA to be compacted into chromosomes as shown above.
We actually have 46 chromosomes in most of our cells, or 23 pairs of chromosomes, as we’ll see below. Each of these chromosomes is a single long piece of DNA wrapped up tightly.
What’s a euploid, aneuploid and mosaic embryo?
The basic idea behind PGT-A is that embryos that have the right number of chromosomes (euploid) have better success than embryos with the wrong number of chromosomes (aneuploid).
We have 46 chromosomes: 23 come from the egg and 23 from the sperm. 22 of these are “autosomes” and are numbered 1-22, and 2 are sex chromosomes (X and Y). These 46 chromosomes give us all the instructions we need for life.
The sex chromosomes determine our biological sex:
- A female has two copies of the X chromosome
- A male has a copy of X and a copy of Y.

Chromosomal aneuploidy is when there’s any number of chromosomes other than 46. So if your embryos has 45 chromosomes, or 49 chromosomes, then the embryo is aneuploid. An embryo with 46 chromosomes has the correct number and this is euploid.
When your embryo is aneuploid, it has a much higher chance of miscarrying or not implanting, as we’ll see below.
So what about mosaics?
A mosaic embryo is an embryo that has a mix of aneuploid and euploid cells, as you can see below:

A mosaic embryo tends to have success rates that are intermediate to a euploid and aneuploid embryo, which we’ll describe more below.
What causes chromosomal aneuploidy?
Aneuploidy is caused by errors during meiosis.
Meiosis is a special type of cell division that produces our sex cells, like the sperm or egg cells. During meiosis the 46 chromosomes that we have in most cells is divided up to 23 chromosomes (1 of each). So an egg cell, and a sperm cell, will each contain 23 chromosomes, and when fertilization occurs these chromosomes get mixed up to give a total of 46 again!
This is why our sex cells contain half the number of chromosomes: so they make the right number (46) after fertilization.
If an error occurs during meiosis, then the egg or sperm cell might have an extra copy of one of the chromosomes.
If an egg cell makes a mistake in meiosis, it might have two copies of chromosome 6 (for example), to give it a total of 24 chromosomes. So when it combines with a sperm cell that has 23 chromosomes it will make an embryo with 24 + 23 = 47 chromosomes, which is aneuploid.
Aneuploid embryos are generally totally aneuploid — all the cells of the embryo are aneuploid. This is because the egg or sperm itself were aneuploid, so all the cells of the embryo inherit this. Mosaic embryos have a mix of aneuploid and normal cells, which are due to errors in mitosis (not meiosis), but we’ll describe that more below.
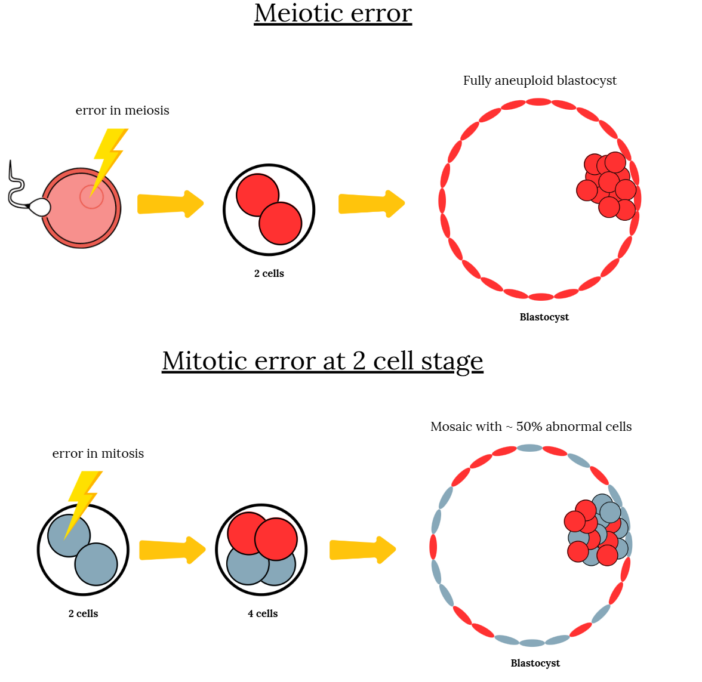
Aneuploidy occurs more frequently with age, particularly in females (as we’ll see in the next section). This may be related to a particular component of the egg cell called the mitochondria.
Mitochondria are specialized components of our cells that produce energy, and as we age they tend to produce less energy. Less energy for the egg cell means that more mistakes might happen during meiosis, such as errors in separating chromosomes.
Read more in my post: Chromosomal errors in IVF: What is aneuploidy and what causes it?
Is aneuploidy attributed more to the egg or the sperm?
Bonus et al. (2022) used single nucleotide polymorphisms (SNPs) to identify parental origin of aneuploidies in embryos that were biopsied. They found that the majority of aneuploidies were of maternal origin:

Interestingly, they didn’t find any association of paternal age with aneuploidy, although the sample size was small. Read more in my post: Paternal (male) effect on embryo aneuploidy examined.
History of PGT-A
The history of PGT-A all starts with the idea that chromosomal aneuploidy is the main reason embryos fail to implant or miscarry. By determining which embryos are euploid, we should have a better chance at choosing the right embryo to transfer.
PGS 1.0 (first generation)
In the 1990’s there was FISH – fluorescent in situ hybridization – but this was only able to screen a few of the 23 chromosomes and was mainly done using cleavage stage embryos.
For the next 20 years or so FISH was the primary method of testing. However, it wasn’t very good at predicting pregnancy outcomes . In fact some studies flat out showed that testing had worse outcomes compared to untested embryos. So PGT-A was discouraged (Brezina et al. 2016)
PGS 2.0 (second generation)
Culturing embryos to the blastocyst stage started becoming more popular in the 2000s and preimplantation genetic testing started to consider the trophectoderm in the blastocyst for biopsy.
The technology itself also improved and now all chromosomes could be analyzed instead of just a few with FISH.
This included SNP, aCGH and qPCR. Also known as PGS 2.0.
These techniques were good for their time but people who were transferring euploid embryos were having miscarriages, which later tested as aneuploid (Maxwell et al. 2016).
Doubt was being raised over PGT-A and concerns over false-positives was becoming a problem as embryos with good potential may be discarded (Munne et al. 2018).
PGS 3.0 (third generation)
By the mid 2010’s, we started realizing that blastocysts may not be 100% euploid or aneuploid, and that there might be a mix of these cells. This is known as embryo mosaicism and might explain why embryos tested as euploid can fail and re-test as aneuploid.
With improved technology (NGS), we were now able to detect mosaicism. This ushered in PGS 3.0 aka PGT-A. At this point, what was considered PGS testing was now rebranded as PGT-A.
Read more in my post: How PGT-A testing has evolved (1990’s to today).
Day 3 or Day 5 embryos for PGT-A?
In the old days, day 3 (cleavage stage) embryo biopsies were more common. These days they’re considered inferior to day 5 (trophectoderm) biopsies for several reasons:
- Reduced viability and implantation rates with day 3 vs day 5 biopsies (Scott et al. 2013).
- Day 3 grading doesn’t correlate well with euploidy like day 5 embryos (Majumdar et al. 2017).
- Higher mosaicism and embryos may self-correct and become normal by day 5 (Barbash-Hazan et al. 2009).
One perk is that day 3 embryos can be biopsied on day 3 and the results can come back in time for a fresh day 5 transfer. However the negatives outweigh this positive and day 5 (trophectoderm) biopsies are now the norm.
Does ICSI need to be performed for PGT-A, or can I do conventional IVF?
Many labs will not allow patients to do conventional IVF for a PGT-A cycle. This is because the sperm that are mixed with the eggs may get stuck to the zona (shell) of the egg. As the fertilized egg develops into an embryo, the sperm are still stuck to the zona, and the worry is that some DNA from the sperm will contaminate the biopsy sample.
A study by Lynch et al. (2022) found that this probably isn’t an issue, after they deliberately contaminated biopsy samples with sperm and couldn’t detect this contamination by PGT-A.
However, they warned that this is only true for tests involving whole genome amplification (WGA). WGA is often performed before DNA sequencing at the PGT-A lab to make multiple copies of the DNA so it’s easier to sequence. The related PGT-M test may not use WGA. Read more in my post: ICSI for PGT-A to prevent sperm contamination may not be necessary.
Recent recommendations from the ASRM encourage ICSI for PGT-M but not PGT-A. Read more in my post: ASRM’s 2024 committee opinion on the use of PGT-A.
How PGT-A works – the embryo biopsy and DNA sequencing
Let’s look a bit at what happens behind the scenes in the IVF lab. If you’re requesting PGT-A, generally these are the steps that are done:
- A tiny hole is lasered into the zona of the embryo on day 3 (assisted hatching).
- By day 5-7, the embryo becomes a blastocyst and cells start to poke out of this hole.
- The cells that are poking out are suctioned and separated using a laser.
- This biopsied piece is stored in the freezer then sent to another lab for PGT-A.
- The biopsied embryo is frozen until results are received and an embryo transfer set up.

See that tiny piece? This is the piece that is tested by PGT-A. We assume that the rest of the embryo has the same makeup. In general, if the embryo is euploid or aneuploid this is mostly true, but less true for mosaics (discussed below).
So what happens after the biopsy piece is submitted to the PGT-A lab? At the PGT-A lab, the technician isolates the DNA from the biopsy piece and sequences it using next-generation sequencing (NGS).
Once sequenced, the PGT-A lab prepares a report that indicates whether or not the biopsy’s DNA had the right number of chromosomes or not.
The results are sent back to the IVF lab where they can notify the patient who may want to transfer the corresponding embryo.
Biopsy samples and the corresponding embryos are numbered. So each tube will be given a particular number that corresponds to a frozen embryo in the freezer. Once the results are received, this particular embryo can be thawed and transferred.
Here’s a video of the PGT-A process explained:
Does a PGT-A biopsy damage the embryo?
During the biopsy, cells are removed from the embryo. At the stage a biopsy is performed, the blastocyst typically has hundreds of cells, and the biopsy removes about 5-10 cells.
This isn’t a huge number, and euploid embryos that have gone through the biopsy process tend to have better success rates compared to unbiopsied embryos, so it’s assumed that any damage the embryo suffers is minimal.
Tiegs et al. (2019) transferred embryos that were biopsied or unbiopsied, without knowing the results of PGT-A (these results were revealed after pregnancy outcomes were known, and this study was meant to address success rates of aneuploid embryos, which I discuss in more detail below).
They found that sustained implantation rates were the same in the biopsied group and the unbiopsied group (53% vs 54%), suggesting the embryos weren’t damaged or harmed in a way that would affect success rates.
There are a number of RCTs (reviewed in the Does PGT-A work? section of this post) that compare untested embryos to euploid embryos and found no difference in outcomes. If the biopsied embryos were damaged they should have a lower chance of success.
Another study by Singh et al. (2019) found that smaller embryos (likely early blastocysts) that didn’t have many cells had dramatically lower pregnancy rates after biopsy. This suggests that biopsy of smaller embryos should be avoided as they may not recover from the process as well as larger blastocysts. In this situation, it might be best for the embryologist to wait an extra day for the embryo to grow more.
However, this was a small study and needs to be repeated to confirm. Read more in my post: Embryo biopsy of non-hatching embryos dramatically reduces IVF success rates.
Oliva et al. (2021) provides some evidence that day 7 blastocysts, fully hatched embryos, poor quality embryos and embryos that had repeat biopsies are more likely to die after they’re thawed. It’s not clear if these embryos are more sensitive to the freeze/thaw process, or to the biopsy process itself. Read more in my post: What causes embryos biopsied for PGT-A to die after thaw?
Understanding PGT-A reports
After the biopsy samples are submitted to a PGT-A lab and processed, the lab will prepare a report with the results. These reports can be a bit confusing, so let’s go over what we might typically see in a report.
Types of chromosomal abnormalities: monosomies, trisomies and segmental abnormalities
There are a variety of chromosomal abnormalities that you might see in an aneuploid embryo:
- A monosomy is a deletion of a whole chromosome
- A trisomy is a duplication/addition of a whole chromosome
- A segmental deletion/addition affects only a segment of a chromosome

Next, let’s look at how these different types of abnormalities can present themselves in a PGT-A report, while visualizing what’s happening with the actual chromosomes.
Let’s first look at an example of a euploid embryo. A euploid embryo has the right number of chromosomes (46, or two pairs of 23 chromosomes). We can see this below with the written result on top and the chromosomes on the bottom. Notice how this embryo is a male because it’s sex chromosomes consist of a copy of X and a copy of Y (XY; a female would be XX).

Next, let’s look at an aneuploid embryo that has monosomy 22. This embryo is missing a copy of chromosome 22.

In the next example, we can see an aneuploid embryo that has monosomy 22 and trisomy 10. This means it’s missing a copy of chromosome 22 and has an extra copy of chromosome 10.

All of these examples include complete chromosomes – either a chromosome is added (trisomy) or deleted (monosomy).
A segmental aneuploid has only a part of a chromosome affected. This might look something like “dup(16)(q23.2-qter)” in a PGT-A report. Let’s break this example down a bit:
- dup(16) means there’s a duplication on chromosome 16
- (q23.2-qter) means that this duplication is for the q23.2-qter region
The “q23.2-qter” is a particular section of the chromosome. Each chromosome has a short arm (“p”) and a long arm (“q”), that can be divided further into bands that are numbered. The “q23.2-qter” is identifying two regions in the chromosome: q23.2 and qter, so q23.2 to qter correspond to this:

So that particular region of the chromosome that corresponds to q23.2-qter is what’s duplicated. So in this embryo, there’s an extra q23.2-qter piece at the end of chromosome 16.
And here’s how that would look (notice the duplication in the green box for chromosome 16):

Understanding mosaic embryo PGT-A reports
Remember, mosaic embryos have a mix of euploid and aneuploid cells. PGT-A labs may report the results differently, but often “[mos]” is used to identify abnormalities that are mosaic.
Let’s look at some examples.
+2 [mos]
+2, -1 [mos]
- The embryo is aneuploid and all cells have trisomy 2
- The embryo is mosaic for cells that have monosomy 1
For the last example, the embryo would generally be considered aneuploid because an aneuploid result overrides a mosaic result. It doesn’t matter that the embryo is mosaic for -1 because it’s also aneuploid and all cells are affected with +2.
+2, -1 [mos], dup(16)(q23.2-qter) [mos]
- The embryo is aneuploid and all cells have trisomy 2
- The embryo is mosaic for cells that have monosomy 1
- The embryo is mosaic for cells that have the segmental dup(16)(q23.2-qter)
For this last one, the embryo doesn’t necessarily have the same cells affected by monosomy 1 and the segmental duplication, it could be different cells.
When we’re looking at mosaics, some cells are affected and some cells aren’t, so often you’ll see a percentage attached to a mosaic result. The PGT-A lab may not give the percentage of mosaicism, but they might indicate “low” or “high” levels of mosaicism:
- 20-40% = low level mosaic
- 40-80% = high level mosaic
Not all PGT-A labs classify low and high level mosaics the same, and some may use “moderate” also. Read the fine print on the report if it’s not clear, or contact the lab.
Low level or high level mosaics have different success rates, which we’ll discuss below in the mosaic section.
PGT-A reports and embryos classified as complex, chaotic or inconclusive, no result
Sometimes you’ll see embryos classified as “complex,” such as a complex aneuploid or complex mosaic. A complex aneuploid is an embryo with 3 or more aneuploidies, and a complex mosaic is a embryo with 3 or more mosaic abnormalities.
A chaotic embryo is an embryo that has 6 or more aneuploidies or mosaic abnormalities.
Sometimes the embryo biopsy is done and it’s sent to a PGT-A lab, but the lab can’t perform PGT-A. This is either because the sample’s quality is too low for PGT-A, or there aren’t enough cells to do PGT-A. These biopsies come back as a “no result” or “inconclusive.”
For inconclusive results, a study by Cimadomo et al. (2018) showed that it occurred about 1.5-5% of the time and is dependent on the IVF clinic’s technical ability. Sometimes the sample is not loaded properly and the tube is empty (the sample is very small so this does happen), or the sample degrades.
The only option for no result embryos is to either transfer it as is or to perform another biopsy.
Understanding raw PGT-A data
So how is all this data generated?
The PGT-A reports won’t show you the raw data, but you can sometimes request it from the testing lab. Let’s go through how NGS-based PGT-A works and then go through an example of a typical NGS sequencing profile.
The process starts with a biopsy of several cells from the trophectoderm of a blastocyst, which are then amplified using whole genome amplification (WGA) to produce enough DNA for testing. The human genome is about 3.2 billion base pairs (bp) long. In this technique, the DNA is broken into short fragments, called reads, that are typically around 100 to 300 bp in length. Each read is sequenced and mapped to a reference genome, like matching pieces to a completed puzzle.
To detect whether any regions of DNA are missing or duplicated, the genome is divided into bins, or chunks of DNA that typically span 100 kilobases to 1 megabase (100,000 to 1,000,000 base pairs), depending on the platform. Each read is assigned to a bin, and the software counts how many reads land in each one.
- Too many reads in a bin = possible gain (eg. trisomy)
- Too few reads = possible loss (eg. monosomy)
- Somewhere in between = possible mosaicism
You can think of it like scanning a bookshelf: if one shelf is too full or too empty, it might mean something’s missing or duplicated.
Here’s an example from actual NGS data:

In the NGS plot, each green dot is a bin, and the green line traces the estimated copy number across each chromosome. Most chromosomes cluster around the expected value of 2 (meaning there’s 2 copies of most chromosomes). However, chromosome 7 shows a clear full trisomy (3 copies), while chromosome 12 appears mosaic, with bins rising toward a copy number of 3 (40% mosaic). The X chromosome shows two copies, and the Y chromosome is missing, indicating a likely female (XX) embryo.
Platform differences in NGS-based PGT-A
Not all NGS platforms work the same. In PGT-A, a platform refers to the specific combination of sequencing technology, reagents, software, and algorithms used to perform PGT-A and interpret the results.
Two of the most widely used platforms for PGT-A are Illumina’s MiSeq (VeriSeq) and Thermo Fisher’s Ion ReproSeq. Both are accurate and widely adopted, but they differ in:
- Speed (Ion runs faster)
- Degree of automation (Ion has more built-in calling)
- Resolution for mosaicism (30–80% for Ion vs 20–80% for MiSeq)
- Manual vs automatic data interpretation

Each uses its own proprietary chemistry, sequencing method, and algorithm to interpret chromosomal data and make “calls” on whether an embryo biopsy is euploid, aneuploid or mosaic. Testing labs often choose a platform based on workflow needs, turnaround time, and how much manual versus automated analysis is preferred.
These platform differences partly explain why one lab might call an embryo mosaic while another calls it aneuploid. Differences in read size, bin size, amplification method, and algorithm thresholds all contribute.
Understanding which platform was used, and how it handles mosaicism, can help put your PGT-A report into better context.
PGT-A success rates
Chances of getting a euploid embryo are driven by age, embryo grades and the PGT-A lab itself
Maternal age is the driving factor for determining whether or not an embryo will be euploid. Rubio et al. (2019) looked at 130,000 biopsies by NGS testing (this is the current testing method):

Besides age, other factors may play a role in determining whether or not an embryo is euploid. Giulia et al. (2022) showed that embryos that become blastocyst on day 5 were more likely to be euploid compared to embryos that became blastocyst on day 6 or 7. Read more in my post: Transfer of day 7 embryos a viable option.
Embryo grades also seem to be related to euploidy rates. Kyoung Kim et al. (2019) found that excellent quality embryos had a higher proportion of euploids compared to poor quality embryos (47.82% vs 29.60%).
In an interesting study, Bardos et al. (2022) compared ploidy from biopsies of embryos from donor eggs between 4 different (unidentified) PGT-A labs. The 4 labs had different results, although they should have been similar since donor eggs were used to make the embryos.

They also found differences in live birth rates using embryos deemed as euploid from these labs. Read more in my post: Study finds differences in euploid, live birth rates among PGT-A testing labs.
Study: Aneuploid-only first PGT-A cycle has no impact on future PGT-A outcomes
For those who have a first PGT-A cycle and get nothing but aneuploid embryos, don’t fret! Herlihy et al (2022) found that women with a first PGT-A cycle that was aneuploid-only had no bearing on future PGT-A cycles. Furthermore, euploids obtained from these second cycles performed just as well as euploids that were obtained from a first cycle.
Read more in my post: Aneuploid-only first PGT-A cycle has no impact on future PGT-A outcomes.
Success rates for euploid transfers
Simon et al. (2018) looked at about 650 euploid transfers:
- <35: 66% live birth per transfer
- 35-37: 71%
- 38-40: 60%
- 41-42: 72%
- >42: 52%
- Donor eggs: 65%
Reig et al. (2020) found that live birth rates dropped with age, from roughly 63% for women <35 to 53% for women >42, which you can see below:

Read more in my post: Euploid embryos show reduced implantation potential with advancing maternal age.
The euploid’s grade and day it was biopsied influence PGT-A success rates
Many people believe that once an embryo is diagnosed as euploid then embryo grading doesn’t matter anymore. A number of studies have found that this isn’t the case, and that euploids should be prioritized for transfer based on their grades.
Zhao et al. (2018) looked at euploid embryos in women <35:
- Graded as excellent (AA) or good (AB or BA) = 50.0% and 49.7% live birth
- Average (AC, CA or BB) = 42.3% live birth
- Poor (BC or CB) = 25.0% live birth

Besides the grade, embryos that take longer to develop into blastocysts tend to have lower success rates. Giulia et al. (2022) showed that day 7 euploids had lower live birth rates and higher miscarriage rates compared to day 5 euploids (see below).

Read more in my post: Transfer of day 7 embryos a viable option.
Are poor quality embryos worth biopsying for PGT-A?
To extend on what was presented in the previous section, let’s talk about whether or not biopsying poor quality embryos is worth it.
IVF labs may have eligibility criteria for what they biopsy, and in many cases, they won’t biopsy poor quality embryos. They may be worried that the embryo won’t tolerate the procedure, and this is certainly true for blastocysts that have very few trophectoderm cells. But in general, poor quality embryos can be biopsied to give decent results.
Cimadomo et al. (2019) compared PGT-A outcomes when biopsying poor quality embryos:
- 23.5% were euploid compared to 51.0% for good/fair quality blastocysts.
- 10.9% led to a live birth (vs 44.8%)
- 36.4% resulted in a miscarriage (vs 13.9%)
They also found that older women were more likely to produce poor quality embryos, so in some cases poor quality embryos may be all that someone has! Read more in my post: Poor quality embryos – are they worth PGS testing?
You can also check the previous section to see more statistics for poor quality embryos.
Does rebiopsying, or thawing and biopsying an embryo have an impact on success rates?
This is a question that is often asked, particularly among women who decide after embryos have been frozen to biopsy them, or for those who have had inconclusive or no result after a biopsy.
The data on this topic is lacking, and studies have been back and forth on it.
For embryos that are thawed for biopsy:
- Zhang et al. (2023) found no differences in pregnancy and neonatal outcomes in embryos tested by PGT-A that were frozen and thawed once or twice. Read more in my post: Multiple embryo freeze/thaw for PGT-A doesn’t affect pregnancy or neonatal outcomes.
- Aluko et al. (2021) found lower pregnancy and live birth rates in women who thawed frozen embryos for biopsy. Read more in my post: Repeated freezing and thawing impacts IVF outcomes in PGT-A cycles.
For embryos that are rebiopsied:
- In a small study, Bradley et al. (2017) found a reduction in embryo survival (from 98% to 93%) and a reduction in live birth rate (50% to 27%).
- Another small study found no difference in survival or live birth rates with rebiopsied embryos (Cimadomo et al. 2018).
A 2024 meta-analysis combined the results of 10 studies to show that embryos that are thawed for PGT-A, or rebiopsied after inconclusive results, have lower chances of pregnancy and live birth, with higher miscarriage rates. However, this meta-analysis was based largely on lower quality data, so more studies are needed. Read more in my post: Is it safe to thaw embryos for PGT-A, or rebiopsy? Results of a meta-analysis.
Mosaic embryo success rates
See the section on mosaic embryos below!
Aneuploid embryo success rates
I’ve summarized the most up-to-date data across multiple studies on whole chromosome aneuploids here: Aneuploid embryo success rates after transfer, which suggests live birth rates are very low (~1%) but not zero. Here’s a closer look at some of the studies:
- Tiegs 2020 set up a clinical trial where they transferred untested embryos that were biopsied, and the biopsy results weren’t revealed until after the pregnancy results. Of the 102 aneuploids that ended up being transferred, none of them led to a live birth, and all either failed to implant or miscarried. Read more in my post: IVF outcomes following aneuploid embryo transfer.
- Tise et al. (2025) present a case report describes the healthy birth of euploid twin girls from embryos labeled as aneuploid by PGT-A. Read more in my post: Embryos labeled aneuploid by PGT-A lead to healthy euploid twins.
For segmental aneuploids, where only a part of the chromosome is affected, success rates are more encouraging:
- Besser et al. (2024) reported on live births after segmental aneuploid transfers, finding that they have similar rates as high level mosaic segmental aneuploids (about 24%). Read more in my post: Segmental aneuploid embryos after PGT-A lead to healthy live births.
- Tiegs 2020 transferred 39 segmental aneuploids, with a 30.8% rate of sustained implantation/live birth. Read more in my post: IVF outcomes following aneuploid embryo transfer.
How many euploids do I need to guarantee a live birth?
This question is hard to answer because there’s no such thing as a guarantee in IVF. Everyone is different. Some may be lucky and have a live birth after a single transfer, while others may need several euploid transfers for it to work.
A study by Pirtea et al. (2020) found that women who had up to three (single) euploid transfers had a cumulative live birth rate of 92.6%. A follow up to this study was done by Gill et al. (2024), who transferred up to 5 euploids, with a cumulative chance of live birth of 98.1%. This was done in women with recurrent implantation failure. Read more in my post: “True” recurrent implantation failure is rare after five consecutive euploid embryo transfers.
Another study by Xu et al. (2025) suggests that most patients with recurrent pregnancy loss who transfer up to 3 euploid embryos go on to have a live birth. Read more in my post: Most recurrent pregnancy loss patients have live birth after multiple euploid transfers.
This tells us that a euploid embryo will work for most people, but it might take a few tries.
Why do euploid embryos fail to implant or miscarry?
If you read the previous sections, it should be no surprise to you that euploid embryos are not a guarantee. The Pirtea et al. (2020) study found that women who had up to three (single) euploid transfers had a cumulative live birth rate of 92.6%.
So even women with up to 3 euploid transfers may not see a live birth.
The embryo is only one side of all this. The other side is the uterine lining (endometrium). It’s quality, and whether or not it’s receptive, plays a big role in successful and sustained implantation.
PGT-A also isn’t perfect, and can only “see” large chromosomal abnormalities. It’s not sensitive enough for smaller mutations, and if there’s a tiny mutation in a critical gene needed for the embryo to successfully implant, then even a euploid may not make it.
Other reasons include:
- Lifestyle choices
- Infection/endometritis
- Vaginal and endometrial microbiota
- Endometriosis
- Anatomical abnormalities (fibroids, etc.)
- Thrombophilia
- Immunological issues
- The doctor’s ability to perform a transfer
- The lab’s quality
Read more in my post: Why do embryos in IVF fail to implant or miscarry?
Mosaic embryos
Now let’s switch gears a bit and consider mosaic embryos in a bit more detail. I have an entire post dedicated to the topic so if you are looking to expand your mosaic embryo knowledge check out my Complete guide to mosaic embryos.
What are mosaic embryos are how do they form?
So we hopefully understand now that the cells of a euploid embryo are all euploid, while the cells of an aneuploid embryo are all aneuploid. With a mosaic, there’s a mix of cells.

Aneuploid embryos occur due to mistakes in meiosis. The egg, or sperm cell has the wrong number of chromosomes and this gets passed on to all the cells of the embryo. So an aneuploid embryo is completely aneuploid.
Mosaics originate due to errors in mitosis. Mitosis is a different kind of cell division, where a single cell divides to form two identical cells. Mitosis is how an embryo goes from one cell, to two cells, to four cells, and so on. An error at some point (let’s say at the two cell stage), causes some of the cells to be aneuploid, while others aren’t:
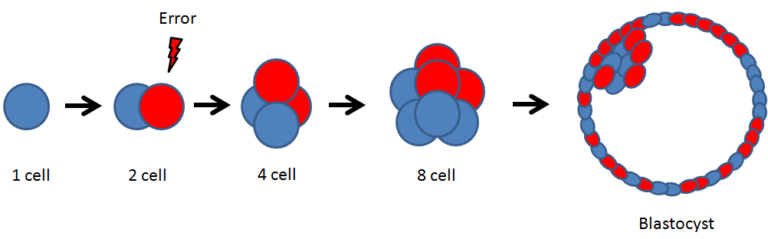
Mosaic embryos can have different amounts of aneuploid cells. Some have 40% aneuploid cells, while others might have 60% or more.
Mosaic embryo success rates
Mosaic embryos can self-correct, a process where euploid cells overtake aneuploid cells, to create a euploid embryo and a healthy baby.
A study by Viotti et al. (2021) compared ongoing pregnancy/live birth rates of 1,000 mosaic embryos that were high level (>50% aneuploid cells) or low level (<50% aneuploid cells). As you can see below, mosaics tend to perform worse than euploids, but still have acceptable success rates.

Read more in my post: Ranking 1,000 mosaic embryo transfers.
Guidelines on transferring mosaic embryos
The 2021 guidelines from the PGDIS (Preimplantation Genetic Diagnosis International Society) recommend mosaics are transferred after considering:
- The level of mosaicism, or % of aneuploid cells present (low level > high level)
- The embryo’s grade (higher quality embryos > lower quality embryos)
- The type of abnormality (whole chromosome > segmental)
They don’t have guidance for specific chromosomes, unlike previous year’s guidance, as the chances of a chromosomally abnormal birth after mosaic transfer are low (based on current evidence).
Read more in my post: PGDIS releases 2021 guidelines for mosaic embryo transfers.
Do I need prenatal testing after transferring a euploid or mosaic embryo?
The 2021 guidelines from the PGDIS recommend prenatal testing for all transfers after PGT-A or IVF in general.
For mosaics with affected chromosomes that could lead to a live birth with a syndrome, it’s particularly important to be sure the prenatal test includes the affected chromosome.
Non-invasive prenatal testing only involves the placental tissue, so the best results will come from an amniocentesis, although there are risks associated with this and it must be discussed with your doctor.
Does PGT-A work?
There’s quite a bit of controversy when it comes to PGT-A, particularly in whether or not the technology actually works in doing what it aims to do: improve live birth rates and decrease miscarriage rates after transferring a euploid embryo. Or is it no different than just transferring a highly graded embryo?
I’ve covered a number of studies on this topic:
- Munné et al. (2019) found no differences in ongoing pregnancy rates or miscarriage rates when transferring a single euploid or a single highly graded embryo. When they broke the data down further, they found that there was an improvement, but only for women 35 and older. I have the full summary on my post PGS testing doesn’t improve success in good prognosis patients.
- Yan et al. (2021) found that women with three or more good quality blastocysts (by conventional grading), had a cumulative live birth rate that was similar to those that transferred up to three euploids. The women were 20-37, and you can check the full summary in my post PGT-A vs embryo grading: results of a 2021 clinical trial.
- A meta-analysis by Chang et al. (2022), combined the results of 7 randomized controlled trials (RCTs) and found no difference in live birth rates among women who used PGT-A or didn’t to select an embryo for transfer. In the same study, a subgroup analysis found that women over the age of 35 who performed PGT-A had improved outcomes, while younger women had a decrease in live birth rates (although it was a small and likely clinically insignificant decrease). The same study found a decrease in miscarriage rates in those using PGT-A. You can read the full summary in my post PGT-A meta-analysis finds live birth rates only improve with older patients.
What does ASRM say about PGT-A? (From my post ASRM’s 2024 committee opinion on the use of PGT-A)
- Universal PGT-A to improve live birth for all patients is unproven
- Evidence is strongest, but still limited, for older women with good ovarian response
- Not supported for routine use in donor egg cycles
- Not enough evidence to recommend routine use for recurrent pregnancy loss
This doesn’t mean PGT-A doesn’t have value — Even though conventional grading may select an embryo just as well as PGT-A in some cases, the primary strength of PGT-A is its ability to identify which embryos are aneuploid so they’re not transferred.
Aneuploid embryos have a very low chance of producing a live birth, so even in younger women PGT-A may be a helpful for preventing their transfer.
What are the miscarriage rates for euploids, mosaics and aneuploids?
I touched on this in the previous section Does PGT-A work?, but but let’s look a bit more at the actual numbers for euploids, mosaics and aneuploids.
The Munné et al. (2019) study found that:
- Overall there was no statistical difference in miscarriage rates (9.9% for euploids vs 9.6% in the untested group).
- For women <35 years old, there was no statistical difference (11.2% for euploids vs 8.3% for untested).
- For women 35-40 years old, there was no statistical difference (8.2% for euploids vs 11.0% for untested).
The Viotti et al. (2021) study looked at the outcomes of 1,000 mosaic embryo transfers:
- Euploid embryos had a 8.6% chance of miscarriage
- <50% mosaic segmental embryos had a 13.6% chance of miscarriage
- >50% mosaic segmental embryos had a 20.3% chance of miscarriage
- <50% one whole chromosome mosaics had a 19.2% chance of miscarriage
- <50% two whole chromosome mosaics had a 11.9% chance of miscarriage
- <50% complex (>2 whole chromosomes) mosaics had a 26.7% chance of miscarriage
- >50% one whole chromosome mosaics had a 31.7% chance of miscarriage
- >50% two whole chromosome mosaics had a 39.9% chance of miscarriage
- >50% complex (>2 whole chromosomes) mosaics had a 44.3% chance of miscarriage
Tiegs (2020) transferred aneuploid embryos:
- 7.2% miscarriage rate for euploids
- 100% miscarriage rate for aneuploids
Read more in my post: PGT-A miscarriage rates (euploid, mosaic, and aneuploid transfers).
How accurate is PGT-A? Does a single embryo biopsy match the rest of the embryo?
People often ask how accurate PGT-A is. If you look at the actual PGT-A report, it usually states something like 98% accurate. This refers to the confidence the PGT-A lab has that the results are true for the biopsy sample submitted.
However, when most people ask about PGT-A accuracy, they mean “how conclusive is a single embryo biopsy?” A typical blastocyst will contain hundreds of cells, so is a 5-10 cell trophectoderm biopsy really representative of the entire embryo? Or with the ICM, the part that becomes the fetus?
In this field of research, the word “concordance” comes up a lot and it means “in agreement with.” So is the first embryo biopsy concordant with the rest of the embryo?
A lot of the information in this section comes from my post Does a PGT-A biopsy match the rest of the embryo? A 2022 update.
A newer 2026 meta-analysis also found higher mismatch rates in segmental aneuploid embryos. Read more in my post: Meta-analysis looks at how well PGT-A biopsies match the embryo.
Euploid, aneuploid and mosaic concordance
Marin et al. (2021) did a review on the topic and included 26 studies made up of euploids, aneuploids and mosaics that were rebiopsied to test for concordance.
Retesting aneuploids found that about 81% were aneuploid after a second biopsy:

Retesting euploids found that about 93% were euploid after a second biopsy:

And retesting mosaics found that about 42% were mosaic:

This shows that aneuploid and euploid embryos are highly concordant. Other studies have also found high concordance rates for euploid and whole chromosome aneuploid embryos, although concordance appears lower for segmental abnormalities and mosaics.
Mosaics, on the other hand, are not very concordant. This may have to do with the nature of a mosaic: it’s a mix of euploid and aneuploid cells. By chance, a biopsy could contain a mix of the cells, or only euploid or aneuploid cells. Check the diagram below to make sense of this:

So overall, aneuploid and euploid results tend to be “accurate” and be representative of the rest of the embryo, while mosaic results are not.
These studies are based off of analyzing a single repeat biopsy (or more in some cases). Other studies have used single cell sequencing, where each cell of an embryo are PGT-A analyzed (and not just a clump of cells from a biopsy). These studies have found that nearly all embryos are mosaic to some degree, with one study suggesting that standard PGT-A could mask mosaic results because of a phenomenon known as “complementary aneuploidy.” Read more in my posts: More sensitive PGT-A test shows that nearly all blastocysts, fetal tissues are mosaic and Mosaicism much more common than previously thought, using more sensitive PGT-A technique.
It’s possible that some proportion of aneuploid embryos are actually high level mosaics (and likewise, some proportion of euploid embryos are actually low level mosaics). What’s desperately needed are more studies sequencing all the cells in embryos, especially aneuploid embryos to see how often they are truly (and fully) aneuploid.
Segmental aneuploid and chaotic embryo concordance
So what about segmental aneuploids? These are actually a bit weird.
Grkovic et al. (2022) rebiopsied 82 segmental aneuploids and found that 40.2% of them were euploid, and therefore reclassified as mosaic. This might be handy for people considering their segmental aneuploids, because as an aneuploid doctors may hesitate to transfer it, but they’d likely have less of a problem if it were a mosaic. Read more in my post: Segmental aneuploids: the main source for PGT-A false positives?
Similarly, embryos that test as chaotic, and have 6 or more abnormalities, tend to have different results the second time around, as shown in a study from Rabkina et al. (2022):

Read more in my post: Preliminary studies examine the rebiopsy and transfer of chaotic embryos by PGT-A.
Does a trophectoderm biopsy match the ICM?
As we’ve seen, embryo biopsies are taken from the trophectoderm, which goes on to form the placenta. The inner cell mass, or ICM, goes on the form the fetus and it’s this part we’re really concerned with.
Capalbo et al. (2021) found that euploid and aneuploid initial biopsy results were highly concordant with a separate ICM biopsy, but less so for mosaics.
How do the overall PGT-A results change with a second biopsy?
It’s important to recognize that a second biopsy result doesn’t erase the first result. Let’s look at a couple of examples.
Let’s say a first biopsy gives the result of aneuploidy for monosomy 2, while the second result is euploid. The overall result is mosaic, because the two tests have shown that there’s a mix of aneuploid and euploid cells:
- Biopsy 1: aneuploid for monosomy 2
- Biopsy 2: euploid
- Overall result: mosaic for monosomy 2
We can apply the same logic to rebiopsying a mosaic:
- Biopsy 1: mosaic for trisomy 21
- Biopsy 2: mosaic for monosomy 1
- Overall result: mosaic for both trisomy 21 and monosomy 1
Do I need PGT-A?
Based on ASRM’s 2024 recommendations, they concluded that PGT-A as a universal screening tool to improve pregnancy rates or reduce miscarriage for all patients is unproven. For other situations:
- Can be considered for women over 35 with a good ovarian response and multiple blastocysts
- Unclear benefit in young, good-prognosis patients
- Evidence doesn’t support routine use for donor egg cycles
- Evidence doesn’t support routine use for advanced paternal age
- Lack of data for benefit in recurrent pregnancy loss patients
Read more in my post: ASRM’s 2024 committee opinion on the use of PGT-A.
That said, the main advantage of PGT-A is identifying aneuploid embryos that nearly always result in failed implantation or miscarriage, and this is true regardless of someone’s age or situation.
Let’s look at some other factors that go into deciding whether to do PGT-A:
PGT-A requires eligible blastocysts, so poor prognosis women who can’t make blastocysts or blastocysts of sufficient quality for a biopsy may not benefit. Ask your clinic what embryo quality is required for biopsy. In many labs, they won’t use poor quality embryos for PGT-A, or won’t take them to day 7 during culture. This can limit the number of embryos available for PGT-A.
PGT-A outcomes are influenced by female age. Younger women are more likely to produce euploid embryos, while the proportion of aneuploid embryos increases with advancing maternal age. As a result, older women may require more cycles to obtain a euploid embryo.
PGT-A is a selection tool, like embryo grading. It doesn’t improve embryo quality or make an IVF cycle more successful on its own. If you don’t have a lot of embryos and would ultimately transfer them all anyway, PGT-A may not be a good choice.
PGT-A may limit your ability to transfer all your embryos. Even though aneuploids rarely work, many women still consider transferring them, and it might be challenging to find a doctor willing to transfer them. Some doctors might also be unwilling to transfer a mosaic, so it’s important to know what your clinic is comfortable doing.
PGT-A technology has limitations:
- PGT-A is based on a 5–10 cell biopsy from the trophectoderm of a much larger embryo, and it’s possible that this biopsy doesn’t represent the whole embryo. While euploid and aneuploid results are usually the same on repeat testing, results are less consistent with mosaics.
- The biopsy is also taken from the trophectoderm, which forms the placenta, not the ICM that becomes the fetus.
- PGT-A detects large chromosomal changes and may not identify smaller genetic abnormalities.
- Some embryos receive an inconclusive or “no result” after PGT-A, and they might need to be rebiopsied. Some studies suggest this can lower success rates
- PGT-A results can also vary between labs depending on the sequencing platform used, how mosaic cutoffs are defined, and how the lab’s algorithms interpret borderline findings..
PGT-A is expensive. PGT-A is an added cost on top of an already expensive IVF cycle. Biopsy and genetic testing are usually billed separately. For some patients, reducing the chance of transferring an aneuploid embryo may justify the expense, but for others, especially those with only a few embryos, the added cost may not change the overall plan.
Euploid embryos are not a guarantee. Euploids may fail to implant or miscarry, with some studies showing live birth rates around 50% — meaning that the other 50% fail to implant or miscarry. Implantation depends on the uterine lining and other factors that PGT-A doesn’t address. Success rates also depend on the grade of the embryo and what day it was biopsied.
Conclusions
PGT-A is a selection tool used to choose embryos that have the right number of chromosomes for transfer. It involves taking a small 5-10 cell biopsy of a blastocyst and sequencing its DNA. Embryos can test as euploid, aneuploid or mosaic.
Euploids have the right number of chromosomes and tend to have the highest success rates, aneuploids nearly always result in failed implantation or a miscarriage, and mosaics have intermediate success rates.
Repeat biopsies of euploids and aneuploids show that these embryos tend to give consistent results, whereas mosaic embryos can give variable results after rebiopsy.
Older women (>35) tend to benefit the most from PGT-A, as they are less likely to produce euploid embryos compared to younger women. Euploid embryos are not a guarantee and their success rates depend on a number of factors, such as the embryo’s grade, the day it was frozen (day 5-7), and the quality of the endometrium (uterine lining).
Whether or not PGT-A actually works for younger women is controversial, and recent data suggests that women <35 have comparable success rates when transferring either a euploid or a highly graded embryo.
If you liked this post and want to support what I do, please consider a paid subscription, Patreon or donate through PayPal!

About Embryoman
Embryoman (Sean Lauber) is a former embryologist and the founder of Remembryo, an IVF research and fertility education website. After working in an IVF lab in the US, he returned to Canada and now focuses on making fertility research more accessible. He holds a Master’s in Immunology and launched Remembryo in 2018 to help patients and professionals make sense of IVF research. Sean shares weekly study updates on Facebook, Instagram, and Reddit regularly. He also answers questions on Reddit or in his private Facebook group.










