Can you actually improve egg quality for IVF? It’s a question many patients ask, and while age is a major factor, it’s not the only one. This guide breaks down what egg quality really means, what affects it (including stimulation, lifestyle, and age), and what the latest research says about supplements and interventions.
🔗 Original studies are referenced in this post or within the linked Remembryo posts.
💡 Reminder: Terms underlined with a dotted black line are linked to glossary entries. Clicking these does not count toward your paywall limit.
Table of Contents
What is egg quality?
What makes a “good” egg or oocyte?
Eggs generally aren’t graded the way embryos are, so defining quality can be tricky. The morphology (looks) of an egg is one way that we can think of quality, but this doesn’t always correlate with success rates.
Besides an egg that looks healthy under the microscope, good egg quality refers to an egg that:
- Can be fertilized
- Can go on to form a good quality embryo, or an embryo that is euploid
- Can lead to a pregnancy or a live birth
Essentially, a good quality egg is one that can go on to form a competent embryo that’s able to lead to a live birth. Egg quality can be inferred by rates for fertilization, blastocyst conversion, euploidy, pregnancy, miscarriage, live birth and so on.
Of course, the sperm also plays an important role in all of these factors. This is why defining egg quality is tricky: because many of the markers of good egg quality are shared with the sperm!
There are no specific tests that can be used to measure egg quality reliably. There are too many factors that can cause things to go wrong with the egg (and sperm!).
Some of the factors that affect egg quality, or the egg’s ability to form a competent embryo that can lead to a live birth, include errors in embryonic genome activation, mutations in maternal effect genes or other genes, aneuploidy, mitochondrial dysfunction, and levels of reactive oxygen species. In fact, these are the same kinds of factors that can lead to an embryo stopping its development (embryo arrest). You can learn more about this in my post Embryo arrest.
How follicles and eggs develop
Follicle development
Eggs come from follicles in the ovaries! As the follicle develops, so does egg inside it. First let’s look at how follicles develop, and then we’ll look at how eggs develop.
The development of a follicle, or folliculogenesis, involves multiple stages (McGee 2000). The first stage is the primordial follicle, which then grows and becomes a primary follicle, then a secondary follicle, then an antral follicle and then a dominant (or Graafian, or preovulatory) follicle.
The dominant/preovulatory follicle contains a mature egg that is released by ovulation.
The follicle, now without an egg, then becomes the corpus luteum and secretes hormones like progesterone to support pregnancy.
You can see this below:

You are born with all the eggs you’ll have. These eggs are contained in very basic follicles called “primordial follicles” and represent your ovarian reserve. Specialized cells called granulosa cells produce Anti-Müllerian hormone, or AMH, which is used to measure the ovarian reserve!
When you were at 20 weeks gestation, you had about 6-7 million primordial follicles, and by birth this was reduced to about 300-400k.
This reduction is caused by a process called initial recruitment, which continues until puberty to further reduce the number of primordial follicles. It’s made up of several stages:
- Some of the primordial follicles go on and develop into “primary follicles”
- These primary follicles go on to develop into “secondary follicles”
- These secondary follicles die (ie. they become atretic)
This process continues until puberty, when you have about 200k primordial follicles left, and your body begins to produce hormones. Lots of them!
In response to these hormones, secondary follicles (from initial recruitment) can develop into “antral follicles,” which can then go on to produce a dominant/preovulatory follicle containing a mature egg for ovulation. This happens every month and is called cyclic recruitment.
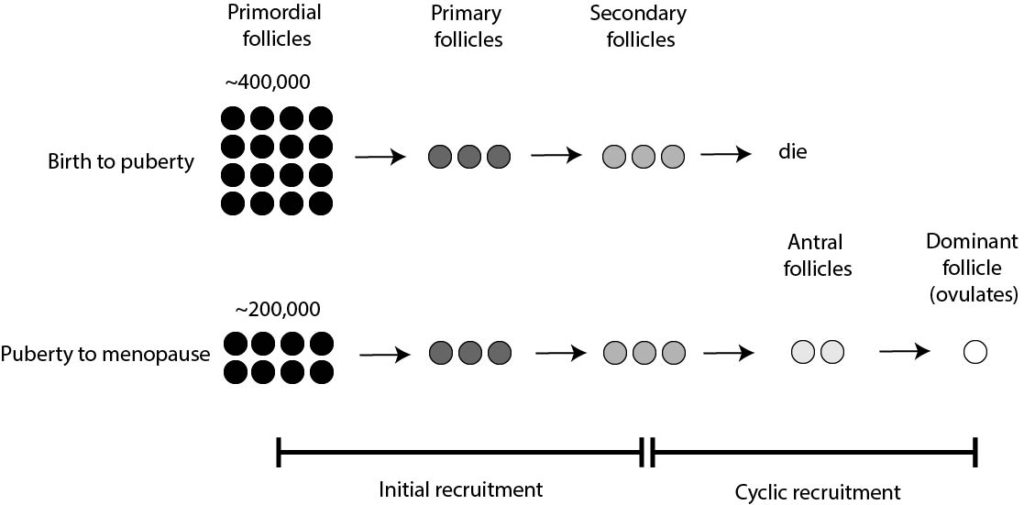
In terms of timing (McGee 2000):
- Primordial follicle to primary follicle: unknown
- Primary follicle to secondary follicle: at least 120 days
- Secondary follicle to antral follicle: 71 days
- Antral follicle to dominant follicle (ovulates): 14 days
Summing this up: 120+71+14 =205 days (plus however long it took for the primordial follicle to make the primary follicle).
As stated by the authors, “the entire growth phase of a follicle is much greater than 220 days or eight menstrual cycles.” In short, egg development takes awhile!
This means that a pool of follicles is selected months before menstruation, with only one typically becoming the dominant follicle to be ovulated during the menstrual cycle. Since this process repeats, the ovarian reserve (made up of primordial follicles) decreases continuously with age. When the pool of primordial follicles is low, menopause occurs.
Oocyte development
Now that we’ve seen how follicles develop, let’s turn our attention to how the egg inside a follicle develops.
There are different stages of an egg’s development as it matures.
It goes from GV –> M1 –> M2.
M1 and M2 refer to metaphase 1 or 2, from the first or second round of meiosis. You can read more about meiosis below.
The earliest stage is the germinal vesicle stage (GV), where you can see the nucleus of the egg. After the GV stage, the nucleus is no longer visible and the oocyte is in the M1 stage. Both a GV and M1 egg are immature.
After the M1 stage, the egg enters the M2 stage and a polar body is seen. The presence of a single polar body is how you know an egg is mature, and is ready to be inseminated.

Putting egg and follicle development together with meiosis
This section here is a bit advanced, but explains more about egg development and how GV/M1/M2 fit into follicle development. You can skip it if it’s too confusing.
Meiosis is a special type of cell division that our body uses to make sex cells like eggs and sperm. The whole point of meiosis is to split up the number of chromosomes so an egg and sperm cell can combine to make the full set.
I won’t get into all the details here, but meiosis consists of two rounds of cell division (meiosis I and meiosis II), each with several stages: prophase, metaphase, anaphase, and telophase. Prophase I refers to the prophase stage during meiosis I
Most of the oocytes in the ovary are contained in primordial follicles (described in the previous section). These eggs are paused in prophase I (as a GV egg), until the the trigger shot is given (or the LH surge in natural cycles, Cakmak et al. 2012). Then the egg resumes meiosis I and continues its development by becoming an M1 (metaphase I) and then an M2 (metaphase II). This process takes about 36 hours. The egg is locked into the metaphase II stage as an M2 until it’s fertilized by a sperm.
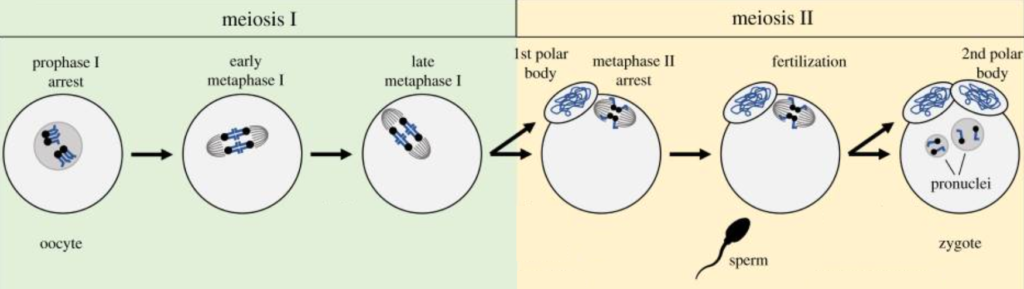
You’ll notice that there’s a polar body during the metaphase II stage. After meiosis I is finished, half of the chromosomes are separated to reduce the chromosome number, and these were placed in a polar body. The polar body is essentially a trash bin to dump excess chromosomes.
A second polar body is released at the end of meiosis II, which happens after the oocyte is inseminated and meiosis II resumes.
Egg morphology
We can’t talk about egg quality without talking about what the egg looks like! The physical appearance of the egg under the microscope is known as its morphology.
Parts of the egg

The oocyte is surrounded by a shell called the zona pellucida (often simply called the “zona”). This protects the oocyte and acts as a container to hold the embryo in as it develops.
Next is the perivitelline space. This is the space between the zona and the egg itself.
Inside the egg is the cytoplasm (or ooplasm) and contains all the mitochondria, amino acids, proteins, and a slew of other factors that the egg needs for its development (and for the development of the early embryo).
A polar body sits outside the egg and this indicates that the egg is mature.
What you can’t see in the above picture is the nucleus. The nucleus of the egg contains the DNA that’s packaged into chromosomes. You can actually see the nucleus as the germinal vesicle at that stage, but not in the stages afterwards.
Common oocyte abnormalities
There are lots of abnormalities that an egg can have. Here are some of the common ones:
- Granular cytoplasm (can be throughout or localized)
- Refractile bodies (fatty deposits in the cytoplasm)
- Ovoid (shaped like an oval)
- Smooth endoplasmic reticulum clusters (sERCs – discussed below)
- Vacuoles (fluid filled sacs in the cytoplasm)
- Thick zona
- Fragments in perivitelline space
- Irregular zona (some regions are thicker than others)
- Large perivitelline space
- Fragmented polar body
- Large polar body
- Large oocyte
There’s some examples that you can check out below, along with a degenerating egg. A degenerating egg is one that is dying. Usually these oocytes appear granular throughout, dark and shriveled up.
The development of these abnormalities might be more common with advancing age.
Egg abnormalities may be linked to lower fertilization and quality – but not always! It’s controversial. (Khalili et al. 2005).
Smooth endoplasmic reticulum clusters, or sERCs, are are aggregates of smooth endoplasmic reticulum, mitochondria and dense granules.

Mizobe et al. (2023) found that these sERCs had comparable fertilization and euploidy rates, but were more likely to form blastocysts. Transfer of a small number of blastocysts led to a 60.0% pregnancy rate and no abnormal neonatal outcomes. Data on the sERCs is limited. Check my post IVF outcomes examined in eggs with smooth endoplasmic reticulum clusters to see the full summary of this study.
In another example of an oocyte abnormality, Ferri et al. (2023) reported on a rare perivitelline space defect which ultimately led to the healthy birth of twins. You can read more about this in my post Case report: Eggs with rare perivitelline space defect lead to healthy twins.
Follicle size and egg maturity
Here’s a typical ultrasound showing an ovary with 6 follicles, each containing an egg:
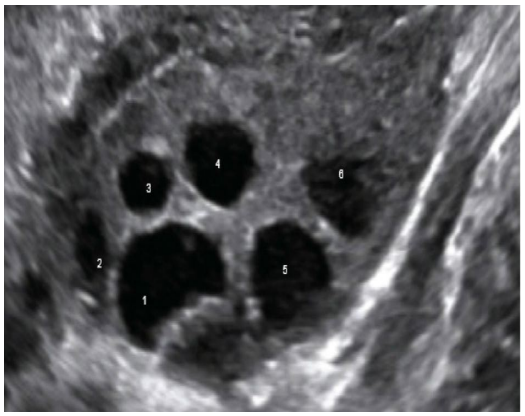
You can see how each follicle is a different size. Follicles increase in size as they develop. Let’s talk about how follicle size relates to egg maturity.
Nuclear maturity vs cytoplasmic maturity
Not all of the eggs obtained during a retrieval will be mature. An immature oocyte is an M1 or GV and can’t be fertilized.
There are two parts of the egg that are involved in maturation:
- The nuclear component – involves the chromosomes/DNA
- The cytoplasmic component – involves the surrounding fluid called the cytoplasm
You can see the cytoplasm below (but you can’t see the egg’s nucleus at this stage).

Both of the nuclear and cytoplasmic components need to mature for fertilization and proper embryo development.
- The nuclear component of the oocyte matures after the trigger shot is given (or after the LH surge in a natural cycle). This triggers meiosis to resume (discussed in the previous section).
- The cytoplasm matures as the oocyte produces and stores special factors in the cytoplasm (this is akin to “ripening”).
It’s easy to tell when the nuclear component has matured by the presence of a polar body in an M2 egg:
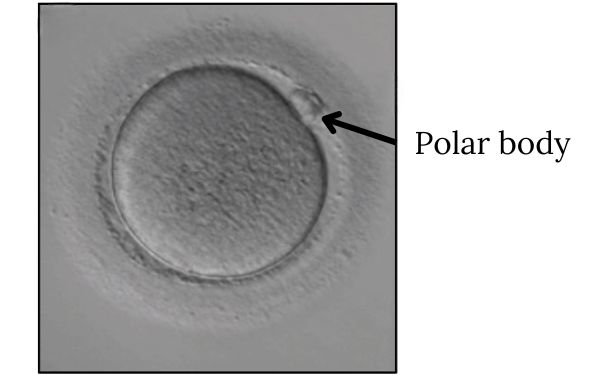
When you hear someone say that an egg is mature – they mean that the egg has a polar body. An immature egg doesn’t have a polar body. The presence of a polar body tells us that the chromosomes have split (one half in the egg, the other half in the polar body) in preparation for pairing with sperm DNA.
Predicting cytoplasmic maturation is more difficult because we can’t see what’s happening inside the cytoplasm.
Follicle size and egg quality
Bigger follicles may be a way of telling that cytoplasmic maturation has occurred!
Shapiro et al. (2022) compared follicle size at the time of trigger and found that larger follicles were more likely to produce mature (M2) eggs:

They also found that larger follicles were more likely to produce good quality embryos, although there was no impact on euploidy rates. You can read my summary of this study in my post Follicle size linked to egg maturity, good quality embryo rate.
McCulloh et al. (2020) also found similar results to this study, with larger follicles producing better quality embryos. You can check out my summary of this study in my post Study compares IVF outcomes by follicle size.
So eggs from bigger follicles tend to be better quality! Follicle size may be one way to predict cytoplasmic maturity.
When follicles get too large (>23 mm) this can lead to a post-mature oocyte. Embryos from these follicles can show reduced quality (Rosen et al. 2008) and are more likely to degenerate after ICSI or not fertilize (personal observation).
There’s some evidence that triggering at a smaller follicle size in older women (the HIER protocol), together with rescue IVM (discussed below), might lead to more transferrable embryos. This might be because the follicle’s metabolism speeds up with age, resulting in more cytoplasmically mature eggs at a smaller follicle size. You can read more about this in my post Early triggering with the “HIER” protocol may be a better approach for older women.
Capper et al. (2022) found that how many mature eggs you get, or your egg maturity ratio, is linked to live birth rates. You can read more about this in my post Low egg maturity ratios linked to reduced live birth rates.
Using rescue IVM to make immature eggs mature
You may have heard of in vitro maturation, or IVM. This is a special type of IVF where immature eggs are deliberately collected, which are then matured in a lab using special culture media and protocols.
A particular type of IVM, called rescue IVM, is a technique that’s used to “rescue” immature eggs from a normal IVF cycle! So instead of discarding any immature eggs, which is typically done, rescue IVM attempts to mature these immature eggs so they can be used.
The process is pretty simple. Eggs are collected as standard, and processed for ICSI. ICSI is the default here because in order to see whether or not an egg is mature, it needs to be stripped of its outer cumulus cells. Stripping is done using an enzyme solution called hyaluronidase, which helps to separate the cumulus cells from the egg. Once the cells are stripped off, you can see which eggs are mature and which aren’t!
Once the oocytes are stripped, the mature eggs can be inseminated as normal, while the immature eggs can be left in the culture media to mature (this is the “in vitro maturation part). Some labs do this for a few hours, while others will wait overnight. Whatever eggs are mature after this period are now “rescued” and can be inseminated!
So how well does rescue IVM work?
Moon et al. (2022) found that M1 eggs that were matured after 2-6 hours had lower fertilization rates, blastocyst conversion rates and euploid rates, but pregnancy and live birth rates were similar. Check out my post Immature eggs matured in-lab for 2-6 h show acceptable IVF outcomes for the full review of this study.
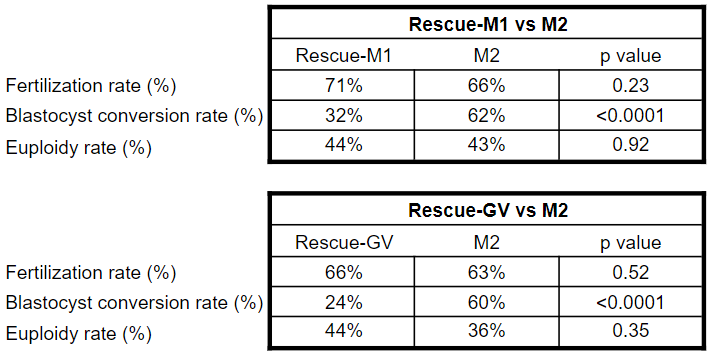
Shani et al. (2023) rescued GV and M1 eggs overnight, many of which became mature and had similar fertilization and euploid rates to M2 eggs from the same cycle, but lower blastocyst conversion rates. You can see this data below and can check out my post Immature eggs cultured overnight by rescue IVM show promising IVF outcomes for more info.
Elkhatib et al. (2023) rescued GV and M1 eggs overnight, which had lower fertilization and blastocyst conversion rates, but similar euploidy rates. You can check my post Culturing immature eggs overnight by rescue IVM increases number of euploids in cycle for a review of this study.
These last two studies were pretty limited in their pregnancy outcomes, since euploids from normal M2 eggs were transferred first. One study also had quite a few double euploid transfers, or mixed transfers with M2 euploids and rescue IVM euploids, making it more challenging to draw conclusions. However, there have been live births and ongoing pregnancies with these transfers.
Overall, it’s something to consider bringing up to your doctor, particularly if you’re getting a lot of immature eggs or not getting many eggs during an IVF cycle.
Advanced maternal age and egg quality
With increasing age, birth rates drop and miscarriage rates rise:

Aneuploidy has been reported in 50-70% of miscarriage products of conception and is considered the leading cause of miscarriages (Hyde et al. 2015). Aneuploidy is the condition of having the incorrect number of chromosomes. For more information on aneuploidy, you can check the beginning sections of my Complete guide to PGT-A.
Maternal age is strongly linked to aneuploidy. In women <35, about 60% of embryos are expected to be euploid, which drops to about 10% by age 44 (Demko et al. 2016).
Aneuploidy results when the oocyte makes a mistake in separating the chromosomes during meiosis, causing some eggs to have too many (or too few) chromosomes. The failure of the egg/sperm to separate its chromosomes in meiosis is known as nondisjunction.
Nondisjunction can occur a number of ways that are age-related:
Let’s look this in more detail! And for even more detail, check out my post Chromosomal errors in IVF: What is aneuploidy and what causes it?
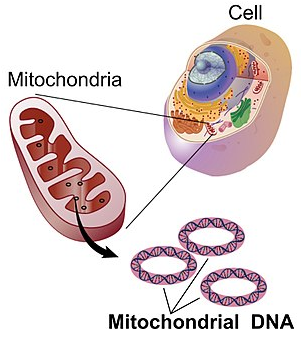
Mitochondrial dysfunction
In order for chromosomes to split up during meiosis, the cell needs energy to power the cellular machinery to get this job done. This energy comes from cellular organs called mitochondria. These are like the batteries of the cell and supply energy when needed. They have their own DNA, called mitochondrial DNA, which provides instructions for mitochondrial components.
Remember how eggs start off in primordial follicles? These follicles represent the ovarian reserve, and they’re with you since before you were even born.
Well, these eggs have mitochondria inside them, waiting to supply energy to the egg once meiosis resumes. And as you get older, the mitochondria get older too.
Over time, they accumulate mutations in their DNA and sometimes those mutations occur in critical genes that are needed for proper mitochondrial function. This can result in the mitochondria not producing enough energy for the cell (Eichenlaub-Ritter et al. 2011, Cimadomo et al. 2018).
Without sufficient energy, the cell might make mistakes during meiosis so it doesn’t split up the chromosomes properly. This can lead to aneuploidy in the egg.
These mutations can occur naturally, or can be influenced by lifestyle choices or environmental factors. The point is, they accumulate over time, causing the mitochondria to become dysfunctional.
You’ve probably heard of CoQ10 as a common supplement to improve oocyte quality. The supplement itself is actually a component of the mitochondria and is needed to make energy. Some cases of mitochondrial dysfunction may benefit from CoQ10 supplementation.
Cohesin dysfunction
Cohesin is a protein “glue” that sticks chromosomes together that can weaken with age.
During meiosis, chromosomes are doubled and stuck together using cohesin. Then they’re split apart to make egg or sperm cells.
The problem is, as you get older, there’s less cohesins to work with (Tsutsumi et al. 2014) so the cohesin glue isn’t as strong and this can result in chromosomes being split apart unequally, leading to aneuploidy.
Spindle dysfunction
The spindle is a cellular machine that has the job of separating the chromosomes during meiosis. The spindle produces fibers that anchor onto chromosomes and pull them to opposite sides of the cell. By separating the chromosomes to opposite sides, the cell can then divide into two, with each cell containing half the chromosomes.
You can see this below, going from one cell in A to two in D. Notice how the chromosomes are being separated by the fibers from the spindle.

There’s a safety checkpoint that the cell uses to make sure chromosomes are split apart normally called the spindle assembly checkpoint or SAC. In older women, the SAC is compromised (Cimadomo et al. 2018) which can result in higher errors in separating chromosomes, leading to aneuploidy.
For more details, check out my post Chromosomal errors in IVF: What is aneuploidy and what causes it?
Genetics and egg quality
There are a number of genes that can influence egg quality and lead to embryo arrest. Embryo arrest is when an embryo stops developing, and this usually occurs around day 3 or so of the embryo’s development.
Why day 3? This has to do with the “genome activation” stage of an embryo’s development.
After fertilization, the sperm’s and egg’s DNA combine to form the embryos DNA, or its genome. Genome is the word we use to describe the complete set of DNA of an organism. It contains all the instructions for the embryo to produce the factors it needs to divide its cells and develop into a human.
The embryo’s genome isn’t turned on until about day 3.
Before this period, the egg relies on stored factors inside the egg to drive cell division. If these factors are compromised, then the embryo might not be able to activate its genome and could arrest.
These factors are mostly coded by “maternal effect genes” in the egg, and errors in them can lead to embryo arrest.
This is discussed in a lot more detail in my post Embryo arrest.
Ovarian stimulation and egg quality
While only one egg is typically ovulated during the menstrual cycle, the goal of ovarian stimulation in IVF is to retrieve many eggs. This involves the use of various medications, usually including gonadotropins like FSH, to maximize the number of follicles that grow to mature an egg.
A meta-analysis by Datta et al. (2021) combined the results of 31 RCTs that compared mild (≤150 IU gonadotropin daily dose) vs conventional ovarian stimulation (>150 IU) in poor, normal and high responders. They found:
- No differences in live births, cumulative live births, ongoing pregnancy rates and clinical pregnancy rates.
- Mild ovarian stimulation resulted in fewer eggs retrieved and fewer embryos.
- No difference in high-grade embryos.
Irani et al. (2020) wanted to see if ovarian stimulation with high amounts of gonadotropins resulted in decreased egg quality (by measuring aneuploidy in the resulting embryos), or decreased live births. They found no differences in aneuploidy or live birth rates in terms of duration of treatment or gonadotropin dosage. You can check out more details on this study in my post Study examines impact of ovarian stimulation on egg quality.
Another study by Hong et al. (2019) compared aneuploidy rates between natural (unstimulated) and gonadotropin-stimulated cycles and found no difference. They also found no difference in sustained implantation rates (beyond 8 weeks).
Egg freezing and egg quality
Does freezing eggs reduce egg quality?
Eggs and embryos are frozen using a technique called vitrification, where the egg is frozen very quickly to -196*C in liquid nitrogen.
A good way to see if oocyte freezing affects embryo quality is to see if there are differences in pregnancy outcomes. A study by Gursu et al (2022) found no difference in pregnancy outcomes when using fresh or frozen donor eggs.

They also found no differences in blastocyst conversion – the number of embryos that went on to form blastocysts. This is a good sign for egg quality!
You can read this study in detail in my post No differences in IVF outcomes when using fresh or frozen donor eggs.
An older study by Forman et al. 2012 compared vitrified eggs vs fresh eggs, and found that there was reduced fertilization and lower embryo quality, but there was no difference in ongoing pregnancy rates or aneuploidy.
Not all eggs will survive the thawing process, but the benchmark for oocyte survival is >80% (Alpha scientists in reproductive medicine, 2012). One study by Cobo et al. (2016) found that age had a strong impact on egg thaw survival:
- <29: 94.5% of eggs survived
- 30-34: 96.1%
- 35-39: 85.8%
- >40: 81.3%
So how well eggs survive may be related to their quality to begin with.
For more information on egg freezing, check my post Egg freezing and thawing.
Diagnosis and egg quality
Diminished Ovarian Reserve (DOR)
Diminished ovarian reserve (DOR) is a reduction in the quantity of the ovarian reserve (primordial follicles) and is usually diagnosed by (Rasool et al. 2017, Jirge 2016):
- Advanced age (>40)
- Poor ovarian response to stimulation (<3 oocytes)
- AMH 0.5 – 1.1 ng/ml, antral follicle count 5-7
In women with DOR the primordial follicle pool is reduced so less follicles can be recruited so there are fewer potential eggs available.
DOR doesn’t affect quality so much as quantity.
Women with large numbers of eggs retrieved have more chances to make good embryos because they have more eggs. Let’s say 25% of eggs retrieved will go on to make a good quality embryo. For someone with 20 eggs that means 5 embryos will be good quality, but for someone with only 4 eggs, there’s only 1 good quality embryo.
Having a low AMH means fewer eggs are retrieved, which can result in higher cycle cancellations.
Morcel et al. (2022) compared outcomes for women with very low AMH levels. They found that more than half of cycles were cancelled in women with AMH <0.4 ng/ml, as a result of lower egg numbers:

However, they didn’t find any changes in clinical pregnancy and live birth rates, suggesting that the quality of the eggs wasn’t affected. You can check out my post Comparing IVF outcomes from cycles with low AMH to read more about this study.
Younger women can also have low AMH and it isn’t only a problem for those with advanced age. Another study by Romanski et al. (2022) compared women with very low AMH (≤0.3 ng/ml) across different ages. They found that these women had a low number of oocytes retrieved that was more pronounced with older age:

They also found that women with very low AMH needed to have 3 IVF cycles to have the same live birth rates as women with normal AMH. Because there were fewer usable embryos per cycle, it takes more cycles to get more embryos and more chances at a transfer. So this doesn’t necessarily indicate a problem with quality.
You can read the full summary of this study in my post IVF and pregnancy outcomes in women with low AMH (≤0.3 ng/ml), by age.
Endometriosis
Endometriosis is a very common disease affecting upwards of 50% of infertile women (Giudice 2010) and is a condition where endometrial-like cells are found growing outside of the uterus.
This may happen during a menstrual cycle where menstrual contents (including endometrial cells) can flow back up through the Fallopian tube and end up in places they shouldn’t be.
This can form an inflammatory reaction that can make sticky adhesions that can fuse different tissues together and distort the anatomy to cause fertility problems (Giudice 2010).
The inflammation associated with endometriosis can create an environment of chemical signals that can be harmful to egg quality. It can also be quite painful.
In a review by Latif et al. (2023), the authors note a number of studies that link egg quality to endometriosis, in terms of:
- Reduced fertilization rates
- Reduced potential for maturity during IVM
- Increased abnormalities of oocyte morphology
- Reduced implantation, clinical pregnancy and live birth rates
However, a number of studies have also shown no impact, with no differences in fertilization, implantation/pregnancy/live birth rates, euploidy rates and number of top-quality embryos. So the research is conflicting.
A recent meta-analysis by Qu et al. (2022) combined the results of 70 studies relating to endometriosis and IVF. Some of their findings include:
- No difference in live birth rates.
- Lower implantation rates with endometriosis.
- Fewer eggs retrieved with endometriosis.
Qu et al. (2023) found the women with endometriosis were more likely to produce aneuploid embryos, suggesting that endometriosis might decrease egg quality, however there was no difference in live births after transferring a euploid. You can read more about this in my post Patients with or without endometriosis have similar live birth rates after PGT-A.
They did find an increase with certain obstetric and perinatal risks, leading the authors to conclude that pregnant patients should be monitored more closely. You can read the full summary in my post Meta-analysis compares impact of endometriosis on IVF, obstetric and perinatal outcomes.
Another related condition is adenomyosis, which involves the presence of endometrial-like cells in the muscular wall of the uterus. A study by Bourdon et al. (2022) found that women with severe endometriosis, combined with adenomyosis, had reduced live birth rates. You can check my full summary in my post Adenomyosis by MRI reduces live births in endometriosis patients.
Polycystic Ovarian Syndrome (PCOS)
PCOS is typically diagnosed by (El Hayek et al. 2016):
- Hyperandrogenism (increased production of male sex hormones like testosterone)
- Irregular menstrual cycles
- Polycystic ovaries (as measured by ultrasound) characterized by a high number of small follicles
In PCOS, higher levels of androgens disrupt the hormone balance involved in follicle development. This results in an abundance of smaller, less developed, follicles (Dumesic et al 2008). Many of the follicles often contain immature eggs.
Infertility is about 10 times more common in women with PCOS compared to those without it (Hart et al. 2015).
Women with PCOS are more likely to experience OHSS, so freezing embryos is often recommended over fresh transfers (Zhang et al. (2022)).
Because women with PCOS often have irregular menstrual cycles, natural cycles for frozen embryo transfers aren’t recommended. Instead, medicated FETs, or FETs with mild ovarian stimulation (using medications like letrozole and gonadotropins) are often used. Zhang et al. (2022) found improved outcomes when using mild ovarian stimulated FETs over medicated FETs.
Because of the abundance of smaller follicles, they might not develop at the same rate and this could affect quality. Chen et al. (2008) found that women with PCOS had:
- More eggs retrieved but fewer mature oocytes
- Lower fertilization
- Fewer highly graded embryos
Despite this, a meta-analysis by Sha et al. (2019) combined the results of 29 studies and found higher live birth rates for women with PCOS. They also noted an increased risk of miscarriage, OHSS, gestational diabetes, preterm birth, hypertension, and other adverse events.
Lifestyle factors and egg quality
Smoking
In women who smoke, Linsten et al. (2005) found that they had a reduced live birth rate (down by 30%) and increased miscarriage rate (up by 5%). They had about the same chances as having a live birth as someone 10 years older!
Waylen et al. (2009) found that smoking reduced the odds of pregnancy and live birth rate by about 50% but didn’t see a difference in miscarriage or fertilization rates.
Setti et al. (2021) found that smoking was associated with abnormal egg morphology, and reductions in fertilization, blastocyst conversion and live birth rates, with increases in miscarriage rates. You can check out the full summary in my post Oocyte quality and IVF outcomes linked to maternal lifestyle and diet.
Egg quality is likely reduced by mutagens in cigarette smoke. These can find their way into the follicular fluid surrounding the oocyte (Waylen et al. 2009) and increase oxidative stress (Ruder et al. 2008).
Marijuana
With marijuana now legal in many parts of the world, more people are using it. Unfortunately there isn’t much research on the topic of IVF outcomes and female marijuana use.
Nassan et al. (2019) found that women who use marijuana are more likely to experience a pregnancy loss.
Har-Gil et al. (2021) found that couples that used marijuana showed no differences in IVF outcomes, including egg maturity, fertilization, blastocyst conversion and ongoing pregnancy rates. You can read the full summary in my post Small study finds comparable IVF outcomes with marijuana smokers.
While the previous study may be reassuring, Roncero et al. (2020) found associations with marijuana use and low birth weight, shorter birth length and a low head circumference, as well as an association with mood and behavioral disorders including anxiety, depression and ADHD.
Alcohol
Firns et al. (2015) did a review on multiple studies:
- One study found that women who had more than 4 drinks a week had a 16% lower chance of a live birth and a 48% reduction in fertilization
- Another study showed that drinking 1 week before retrieval reduced the number of eggs retrieved by 13% and increased the miscarriage rate
Setti et al. (2021) found that alcohol consumption was associated with abnormal egg morphology, decreased pregnancy and live birth rates, and increased miscarriage rates. You can check out the full summary in my post Oocyte quality and IVF outcomes linked to maternal lifestyle and diet.
Increased oxidative stress from alcohol metabolism may affect egg quality (Ruder et al. 2008).
Diet
There are lots of diets out there with supposed positive effects on IVF outcomes.
The Mediterranean diet is rich in fruits, vegetables, whole grains, nuts, olive oil and low in red meat. It has be shown to have a positive effect on pregnancy rates (Karayiannis et al. 2018; Braga et al. 2015). This diet is rich in unsaturated fatty acids (olive oil, fish, nuts) and antioxidants which may play a role in promoting oocyte quality.
Kermack et al. 2019 used a 6-week “Mediterranean diet” intervention where participants received a daily supplement containing omega-3 fatty acids and vitamin D. They found no differences in fertilization rates or pregnancy/live birth rates. You can check out my summary of this study in my post Mediterranean diet and IVF – a clinical trial.
Sun et al. (2019) found increased fertilization and embryo yield with adherence to the Mediterranean diet.
A meta-analysis by Muffone et al. (2023) found no improvement in live birth rates among 11 studies, however this was inconsistent and they concluded that there isn’t enough evidence to support the Mediterranean diet clinically.
Setti et al. (2021) found that consumption of refined sugar and artificial sweetener were linked to increases in abnormal egg morphology and decreases in pregnancy/live birth rates. Consumption of milk/dairy, legumes/vegetables and fish were associated with a reduced risk of abnormal egg morphology. You can check out the full summary in my post Oocyte quality and IVF outcomes linked to maternal lifestyle and diet.
For caffeine, Lyngsø et al. (2019) found no difference in live birth rates in women who drank varying amounts of coffee and those who didn’t. Check out my full summary Study examines impact of coffee on IVF outcomes for more info.
Sleep
There isn’t very much information relating to sleep quality and IVF outcomes.
Yao et al. (2022) found that women with good quality sleep had a higher fertilization rate compared to women with poor quality sleep. Overall this study found that women who sleep about 7-9 hours a night, go to bed around 10 pm (and wake around 6 am), and had a good quality of sleep, tended to perform best in terms of IVF outcomes. You can check out my full summary here Sleep quality and its impact on IVF outcomes for more info.
Qin et al. (2023) performed a meta-analysis and combined the results of 8 studies relating to infertility and sleep disorders. They found no difference in patients with sleep disturbance or sleep apnea, but there was an increased risk in patients who didn’t go to bed early and wake early.
Environmental factors and egg quality
Endocrine disruptors
Endocrine disruptors have a structure similar to sex hormones, and can bind to endocrine receptors and disrupt hormonal signals. This can cause problems with the normal functioning of the endocrine system.
Here’s a look at the hormone estradiol (green) and the endocrine disruptor bisphenol A (BPA, purple).
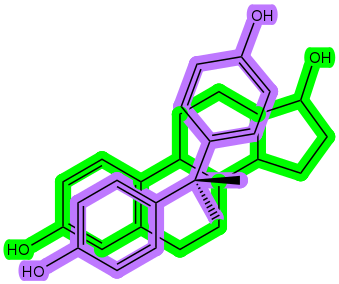
Exposure to endocrine disruptors is through the environment and products involved in everyday life, including plastic materials, canned food and beverages, food, scented products like deodorants and soaps, hygiene products, cosmetics, and much more.
Some common endocrine disruptors are BPA, phthalates, dioxins, organochlorine and organophosphate pesticides, perfluoroalkyl and polyfluoroalkyl substances (PFAS), parabens and phenols.
Numerous studies have demonstrated the detrimental effect of endocrine disruptors on both female and male reproductive health. Most studies show that various interventions can reduce the concentration of these chemicals in the body, but they don’t link this to clinical outcomes. So more research is needed.
I have a detailed post that addresses this topic Endocrine disruptors and their impact on fertility.
Day length and the seasons
Correia et al. (2022) and Leathersich et al. (2023) investigated the association of IVF outcomes following egg retrieval during different seasons.
One study was based in Perth, Australia, while the other was based in Boston, USA. Both studies found an improved odds of live birth when the oocyte retrieval took place in the summer. Because it takes several months for eggs to develop from follicles, this suggests that eggs that start developing in the cooler months of spring may be better quality.
They also found other interesting results. You can check my summy here Studies link season, temperature, day length to egg quality and IVF outcomes.
Improving egg quality
Supplements for egg quality
Supplements can be:
- Vitamins
- Minerals
- Herbs
- Amino acids
- Antioxidants
Most women undergoing IVF take some sort of supplement, usually to improve egg quality. The data with supplements can be inconsistent, and this is due in part to their lack of regulation.
There are a number of problems with the supplement industry that people may not be aware of (reviewed by Starr 2015):
- Traditional drugs need to go through clinical trials to show they work and that they’re safe. This isn’t required for supplements. Supplements are not approved by the FDA and are not regulated like prescription or over the counter drugs.
- Supplements can contain ingredients at any concentration and in any combination, making them very inconsistent. One report found that vitamin D concentrations ranged from 9% to 146% of the stated concentration.
- Reports have found that supplements can contain contaminants, or ingredients not listed on the label. In one report, 83% of herbal products switched the active ingredient to something else.
- Manufacturers set their own standards, and the same ingredient from different manufacturers can be very different in terms of composition, strength or bioavailability.
- Because supplements aren’t regulated, and they’re cheap to make, it’s a huge industry: there were $30 billion in sales in 2011 (this has doubled in 2023). It’s driven by commercial interest and supplements are marketed with misleading language to sell, making people believe that they are just as effective as drugs (stating that they’re “clinically tested” or “doctor formulated”), or can use words like “natural” to make them seem more attractive. An investigation by the Office of Inspector General revealed that most supplement health claims are misleading, uninformative and inconsistent.
A recent Cochrane review (Showell et al. 2020) summarized the data on a variety of supplements for improving egg quality. Below I’ll just share the clinic pregnancy data, but you can check out all the details my post Impact of supplements on live birth and pregnancy rates in women: a meta-analysis.
Type of supplement:
- N-acetyl-cysteine: increased pregnancy rates (8 RCTs, 1,590 women)
- Melatonin: increased pregnancy rates (7 RCTs, 678 women)
- Vitamin E : no difference in pregnancy rates (1 RCT, 103 women)
- Vitamin C : no difference in pregnancy rates (2 RCTs, 899 women)
- L-arginine : no difference in pregnancy rates (2 RCTs, 71 women)
- Vitamin D : no difference in pregnancy rates (1 RCT, 52 women)
- Vitamin B : no difference in pregnancy rates (1 RCT, 102 women)
- Myo-inositol + folic acid : no difference in pregnancy rates (1 RCT, 94 women)
- CoQ10: increased pregnancy rates (4 RCTs, 397 women)
- L-carnitine: increased pregnancy rates (2 RCTs, 450 women)
- Note that N-acetyl-cysteine and L-carnitine both had significant heterogeneity (variability) in their studies, so it’s less clear if these are effective.
- Note that the quality of evidence is indicated as LOW for these studies.
For a lot more details on supplements, check my post Improving egg quality with supplements.
In addition to the supplements above, there are promising new supplements emerging, like spermidine. In a study in mice, spermidine was able to improve egg quality in older mice, by increasing egg maturity rates and decreasing the chance of aneuploidy. However, more work needs to be done to determine what dose would be safe in humans. You can read more in my post Spermidine supplement improves fertility, egg quality in aged mice.
Mitochondrial donation
Mitochondrial donation involves removing the nucleus and DNA from a donor egg, and then transferring the nucleus from a patient’s egg into the enucleated donor egg. Yes, it’s as confusing as it sounds. Here’s a diagram to put it together!

The outcome of this is the patient’s nucleus (and DNA) is now in the donor egg, which has healthy mitochondria. These healthy donor mitochondria might make fewer errors during meiosis than the patient’s mitochondria, potentially leading to improved euploidy rates and better outcomes.
Since the technique involves three individuals — the mother, father and donor — the embryos are sometimes called “three parent embryos,” or “three parent babies” when the baby is born.
There isn’t much research on mitochondrial donation, because it’s illegal in most countries and is considered a type of human genome modification. In countries where it is permitted (the UK and Australia), it’s allowed only for people with mitochondrial disorders and not for people with poor egg quality.
There are certain countries where it isn’t regulated, and people will often travel to these places to have the treatment done (ie. Ukraine, Greece, Albania).
Costa-Borges et al. (2023) performed the first pilot study investigated mitochondrial donation in patients with poor egg quality. They found that all the women in the study had improved fertilization and blastocyst production, with 6 going on to have a live birth. All the babies were healthy.
You can read more about this study and mitochondrial donation in my post Pilot study shows improved IVF outcomes after spindle transfer in women with poor egg quality.
Artificial oocyte activation
Once the sperm penetrates an oocyte, the process of fertilization begins. An important part of fertilization is “oocyte activation.”
As a reminder, meiosis is a special type of cell division that our body uses to make sex cells like eggs and sperm. Mature, or M2 eggs, are locked in a particular phase of meiosis until fertilization. The egg can’t be fertilized until meiosis continues, and this requires energy.
Once the sperm enters an egg, it triggers the release of calcium ions (Ca2+ ions) from the egg. This acts as a switch that turns on the mitochondria in the egg to start producing energy. This energy is used to resume meiosis so fertilization can occur and the zygote can form.
In some cases, poor egg quality can be due to problems in activating the egg. This can cause very low fertilization or total fertilization failure. One way to correct this is to provide more Ca2+ ions to try and initiate oocyte activation artificially (artificial oocyte activation, or AOA).
AOA involves providing more Ca2+ ions to the egg using chemicals called ionophores, which carry Ca2+ ions, or by using chemicals like strontium chloride that can trigger Ca2+ release from the egg.
Fawzy et al. (2018) used AOA, which allowed fertilization to occur, and found improved IVF and pregnancy outcomes in patients with no/low fertilization. You can read more in the post Artificial oocyte activation improves IVF outcomes in clinical trial.
Growth hormone (omnitrope)
Growth hormone (GH) is secreted by the pituitary gland and leads to synthesis of insulin-like growth factor 1 (IGF-1) in the liver. IGF-1 receptors are present on egg cells and the surrounding granulosa and theca cells in the follicle, and can assist in the process of egg maturation. For this reason, GH is often used during ovarian stimulation for poor responders (as Omnitrope) and may improve the quantity and quality of eggs retrieved (Hart 2019).
Shang et al. (2022) performed a meta-analysis and combined the results of 25 RCTs involving the use of GH. They found that women who used GH had increased pregnancy rates and egg maturity rates compared to those who didn’t use GH.
To read more about this study, check my post Meta-analysis shows growth hormone improves endometrial function.
Conclusions
Egg quality relates to the egg’s ability to fertilize, form a blastocyst and lead to a pregnancy and live birth.
The size of the follicle not only relates to the chance of getting a mature egg, but is also involved in the quality of the egg. Immature eggs can be rescued using rescue IVM, which might be a good strategy to increase mature egg numbers.
With advancing age comes poorer quality eggs, which is related to the quality of individual components inside the egg, like the mitochondria or the spindle.
The IVF process itself, including ovarian stimulation and egg freezing/thawing, might have an impact on egg quality, and research is ongoing. Additionally, diagnoses, lifestyle factors and environmental factors can all play a role in egg quality. Some of these factors can be prevented, for example by eating healthier and avoiding exposure to endocrine disruptors.
Various interventions can help improve egg quality, although better research is needed to be confident in these approaches, particularly for supplements. Mitochondrial donation holds promise as an effective way to improve egg quality, but research is lacking due to regulatory concerns. As research continues, and we learn more about this technique, it may become more accepted.
If you liked this post and want to support what I do, please consider a paid subscription, Patreon or donate through PayPal!

About Embryoman
Embryoman (Sean Lauber) is a former embryologist and the founder of Remembryo, an IVF research and fertility education website. After working in an IVF lab in the US, he returned to Canada and now focuses on making fertility research more accessible. He holds a Master’s in Immunology and launched Remembryo in 2018 to help patients and professionals make sense of IVF research. Sean shares weekly study updates on Facebook, Instagram, and Reddit regularly. He also answers questions on Reddit or in his private Facebook group.