This post covers the variety of reasons embryos can fail to implant or miscarry, including as a result of advanced maternal age, chromosomal translocations, lifestyle, endometritis, the microbiota, immunological issues, endometrial receptivity, the clinic, and more. Note that this is not a complete list, and will be updated periodically.
🔗 Original studies are referenced in this post or within the linked Remembryo posts.
💡 Reminder: Terms underlined with a dotted black line are linked to glossary entries. Clicking these does not count toward your paywall limit.
Table of Contents
Implantation failure or miscarriage?
First we’ll need to define what we mean by an embryo “failing”. The embryo can either fail to implant, or it can implant and then miscarry.
Implantation is when there is ultrasonic evidence of an intrauterine gestational sac (at 3-5 weeks). If no gestational sac forms, then it’s considered implantation failure (Coughlan et al. 2013). Implantation failure could occur early on where the embryo doesn’t even attach to the endometrium, or later after it attaches but dies before a gestational sac forms.
A term related to the implantation rate is the clinical pregnancy rate. A clinical pregnancy is a pregnancy that’s detected in someway by ultrasound. So either a gestational sac is detected (3-5 weeks, which is also used to confirm implantation as described above), the yolk sac (around 6 weeks), the fetal pole (around 6.5 weeks), or the fetal heartbeat (around 7 weeks). So it’s fair to say that if a clinical pregnancy did not occur, there is also implantation failure.
Miscarriage is typically defined as pregnancy loss after a clinical pregnancy, usually before 20 weeks gestation.
What’s to blame? The embryo, the uterus, or clinical factors?
In the case of euploid embryos, we might be tempted to assume the embryo is not to blame. In many cases, this may be true! Pirtea et al. (2020) found that about 92% of women who transferred 3 single euploid embryos had a live birth. This tells us that in most cases the embryo quality itself determines whether or not the embryo implants. Read more in my post: “True” recurrent implantation failure is rare after five consecutive euploid embryo transfers.
In contrast, Craciunas et al. (2019) estimate that embryos account for about one-third of implantation failures, while endometrial receptivity and embryo–endometrial interactions account for the rest.
So which is it?
In reality, both matter. Embryo quality appears to be the main driver in most cases, especially due to chromosomal issues. However, once good quality or euploid embryos are used, uterine and other factors may become more important, particularly when failures happen repeatedly. Even then, a euploid embryo is not a guarantee and can still fail.
To make sense of this, we can group the causes of implantation failure and miscarriage into three broad categories:
- Embryo-related factors (such as chromosomal abnormalities)
- Uterine and maternal factors (such as endometrial receptivity and inflammation)
- Lab and clinical factors (such as embryo handling and transfer technique)
These categories can overlap. For example, even a healthy embryo may fail if the uterine environment isn’t optimal, and lab or transfer conditions can also play a role.
Now let’s look at the different reasons embryos may fail to implant or miscarry.
Embryo-related causes of implantation failure and miscarriage
Aneuploidy
Aneuploidy refers to an abnormal number of chromosomes in an embryo, and is one of the leading causes of implantation failure and miscarriage.
Qin et al. (2024), in their study of over 3,200 pregnancy losses, showed that about two-thirds involved a chromosomal abnormality, with most of those being aneuploidies. Read more in my post: Study performs genetic analysis on over 3,200 miscarriages.
Normally, embryos have 46 chromosomes (23 pairs), with one set coming from the egg and the other from the sperm. If there’s an extra or missing chromosome, the embryo is considered aneuploid. A euploid embryo has the right number of chromosomes.
Aneuploidy usually occurs due to errors during the formation of the egg or sperm. These errors happen during a process called meiosis, where chromosomes are separated so that each egg or sperm ends up with 23 chromosomes.
If chromosomes don’t separate properly, the resulting egg or sperm may have too many or too few chromosomes. When fertilization occurs, this leads to an embryo with the wrong number of chromosomes.
Female age is one of the strongest factors linked to aneuploidy. Eggs are formed before birth and remain paused for years, which increases the risk of errors when they resume development later in life. This leads to an increased chance of aneuploidy and is why live birth rates decrease and miscarriage rates increase with female age.

Armstrong et al. (2023) compared PGT-A results after about 85,000 embryo biopsies. The proportion of aneuploid embryos rises from about 23% under 35 to over 75% above 42, showing a steep increase with age.
- <35: 23.4% chance of an aneuploid embryo
- 35-37: 31.3%
- 38-40: 43.3%
- 41-42: 59.8%
- >42: 75.2%
Read more in my post: A look at how PGT-A results change with age, using data from over 86,000 biopsies.
Chromosomal translocations
A translocation is a type of chromosomal rearrangement where a segment of one chromosome breaks off and attaches to another chromosome or a different region of the same chromosome.
In a balanced translocation, no genetic material is gained or lost, so the person carrying it is typically healthy and may not know they have it.
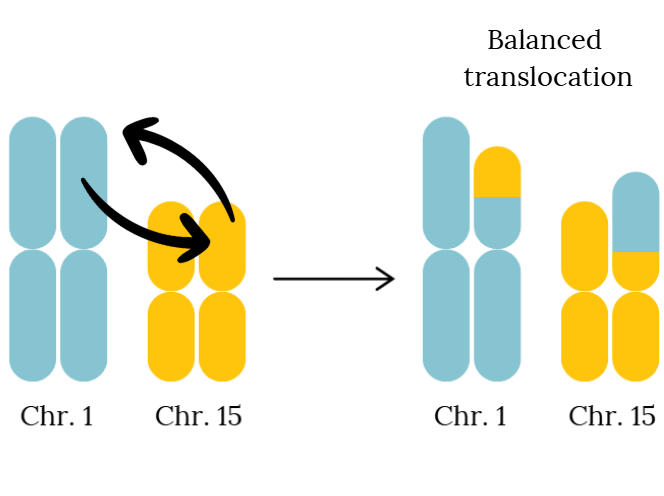
The issue arises during the formation of egg or sperm cells. The rearranged chromosomes can separate unevenly, leading to unbalanced gametes. When these combine during fertilization, the resulting embryo may have missing or extra genetic material, increasing the risk of implantation failure or miscarriage.
Balanced translocations are estimated to occur in about 0.2–0.4% of people (Tsuiko 2019), and are usually diagnosed through karyotyping.
If a translocation is identified, PGT-SR can be used to select embryos that are chromosomally balanced and more likely to result in a successful pregnancy.
In one case presented by Upadhyay et al. (2024), a couple struggling with infertility for 20 years achieved a successful pregnancy after detecting a balanced translocation in the male partner and using PGT-SR. Read more in my post: Karyotyping and PGT-SR help couple achieve live birth after 20 years of infertility.
Embryo quality
An embryo’s quality and development speed can affect whether it implants or leads to a live birth.
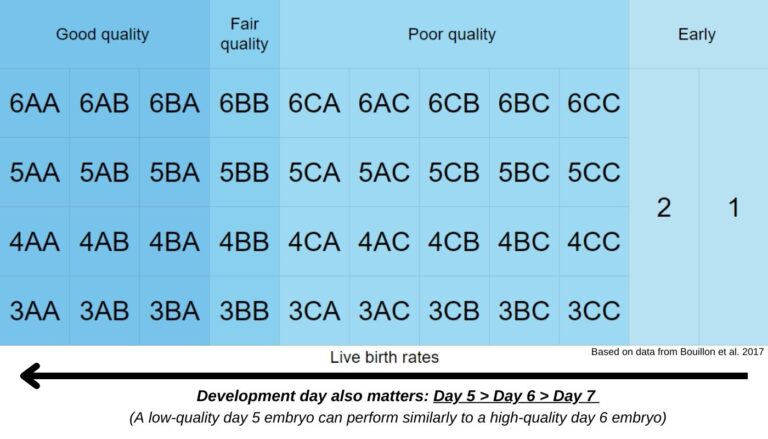
Embryo quality is usually assessed by grading. At the blastocyst stage, this includes:
- The inner cell mass (ICM), which becomes the fetus
- The trophectoderm (TE), which becomes the placenta
- The degree of expansion (how developed the embryo is)
Higher quality embryos tend to have more cells and better organization in both the ICM and trophectoderm, and are more likely to implant compared to lower quality embryos.
But embryo quality isn’t just about appearance. How fast an embryo develops also matters.
Most embryos reach the blastocyst stage by day 5. Those that take longer (day 6 or day 7) generally have lower success rates.
Both embryo quality and development speed affect implantation rates in untested embryos as well as PGT-A tested euploid embryos.
Read more in my post: Complete Guide to Embryo Grading and Success Rates.
Why euploid embryos still fail
Even when an embryo is euploid, implantation and live birth aren’t guaranteed. Many studies put success rates per transfer in the ~50–70% range, meaning failure is still pretty common.
There are several reasons for this:
PGT-A has limitations. It tests only a small sample of cells (about 5–10) from the trophectoderm, not the whole embryo. While results are generally reliable for euploid embryos, that small sample may not fully represent the entire embryo, especially if mosaicism is present.
In fact, more sensitive techniques suggest mosaicism is extremely common, with Chavli et al. (2024) finding it in about 82% of blastocysts. This means many embryos labeled as “euploid” may still contain a small number of abnormal cells that weren’t detected. Read more in my post: Mosaicism much more common than previously thought, using more sensitive PGT-A technique.
PGT-A is a snapshot in time, and an embryo’s chromosomal makeup may change as it continues to develop. Abdelbaki et al. (2025) used real-time imaging to watch embryos divide and found that chromosomal errors can continue to arise at the blastocyst stage. This means that embryos can develop errors even after PGT-A. Read more in my post: Real-time imaging suggests that embryo mosaicism may change over time.
PGT-A can’t detect everything. It’s designed to identify large chromosomal abnormalities, but smaller genetic mutations can still cause pregnancy loss. Arnadottir et al. (2025) estimated that these small mutations may explain about 6% of miscarriages in euploid pregnancies. Read more in my post: Small genetic mutations in euploid pregnancies linked to miscarriage.
Other factors still matter. As mentioned, embryo quality still matters, and the uterine environment and lab conditions also play a role in IVF success. A large meta-analysis by Cimadomo et al. (2023) found that even among euploid embryos, outcomes are influenced by maternal age, embryo quality, repeat freeze/thaw, specific diagnoses, and other clinical or lab factors. Read more in my post: Meta-analysis combines 74 studies to examine factors linked to euploid transfer success.
Putting this together, a “euploid” result doesn’t mean an embryo is perfect. It means it passed one important screening step, but it can still fail to implant or miscarry.
Looking at outcomes over multiple transfers can help put this into perspective. For example, Pirtea et al. (2020) found that transferring three consecutive euploid embryos led to about a 92% chance of live birth. In other words, even if a single transfer fails, the overall chance of success is high with repeated attempts.
Uterine and maternal causes of implantation failure and miscarriage
These factors relate to the uterine environment and maternal health. While embryo quality is often the main driver of success, these factors may become more important when good-quality embryos repeatedly fail.
Infection or endometritis
Endometritis is inflammation of the uterine lining, usually caused by a bacterial infection. This inflammation can disrupt the uterine environment needed for implantation and early pregnancy.
It can be diagnosed using hysteroscopy or by analyzing a biopsy of the endometrial tissue under a microscope.
Endometritis is often asymptomatic, meaning many patients don’t know they have it. It has been reported in up to 45% of women with recurrent implantation failure (RIF) (Lin 2016, Kushnir et al. 2016)
Antibiotics might help in reducing levels of these bacteria and the corresponding inflammation, which can lead to improved pregnancy outcomes. Vitagliano et al. (2018) performed a meta-analysis on IVF outcomes in women with chronic endometritis who were treated and found improved implantation rates (25.2% vs 17.8%), with no change in miscarriage.
More recent research suggests the story may be more complex. Zhang et al. (2023) showed that even after antibiotic treatment, patients may still have an increased risk of pregnancy loss, possibly due to underlying immune or inflammatory changes in the endometrium. Read more in my post: Patients with antibiotic-cured endometritis still show increased pregnancy loss.
Vaginal and endometrial microbiota
The vaginal and endometrial microbiome refers to the bacteria that live in the reproductive tract. These bacteria may play a role in implantation by influencing inflammation, immune signaling, and the overall uterine environment.
In the vagina, Lactobacillus species are typically dominant and are thought to be protective. In the uterus, the microbiome is less well understood, but Lactobacillus is often present along with other bacteria.
Some studies have found that a Lactobacillus-dominant microbiome is associated with better IVF outcomes, while lower levels have been linked to reduced implantation, pregnancy, and live birth rates (Moreno et al. 2016). However, results across studies are inconsistent, and not all research shows a clear relationship.
More recent work suggests that it may not just be the presence of certain bacteria, but what they produce. In one study, women who didn’t become pregnant had a more diverse microbiome with fewer Lactobacillus species, and bacterial metabolites were shown to affect how endometrial cells behave, including inflammation and barrier function. Read more in my post: Researchers examine the microbiome and implantation in unexplained infertility.
There is also interest in testing and modifying the microbiome. Tests like EMMA and ALICE identify bacterial imbalances, and some studies suggest that treating abnormal results with antibiotics or probiotics can improve outcomes. However, the evidence is still limited and not all studies include proper control groups, making it difficult to draw firm conclusions. Read more in my post: Study investigates the use of EMMA & ALICE in IVF patients with RIF, RPL.
Even when probiotics are used, results are mixed. For example, one randomized trial by Thanaboonyawat et al. (2023) found no improvement in pregnancy rates, although miscarriage rates were reduced in some cases. Read more in my post: RCT finds probiotic supplementation in women without RIF may be beneficial.
Overall, the microbiome may play a role in implantation, but its exact impact and how best to modify isn’t clear.
Read more about this topic in my post: The impact of the vaginal and endometrial microbiome on reproductive health and IVF outcomes.
Endometriosis and adenomyosis
Endometriosis involves the growth of endometrial-like tissue outside the uterus, typically in the pelvis (ovaries, fallopian tubes, bladder). This tissue responds to hormones like estrogen, leading to inflammation, bleeding, and the formation of scar tissue (adhesions) that can affect fertility.
A related condition, adenomyosis, occurs when this tissue grows within the muscle of the uterus. Like endometriosis, it’s linked to inflammation and changes in how the uterus functions.
Both conditions are thought to affect fertility through:
- Chronic inflammation and immune changes
- Altered hormone signaling (including progesterone resistance)
- Changes in uterine function and contractility
- Possible effects on implantation timing
For example, Samarajeewa et al. (2025) study suggests the implantation window may be shorter or shifted in endometriosis. Read more in my post: Endometriosis may shorten the window for embryo implantation.
In terms of pregnancy outcomes, the data is mixed:
- A meta-analysis by Qu et al. (2022) found no clear difference in live birth rates, but slightly lower implantation rates. Read more in my post: Meta-analysis compares impact of endometriosis on IVF, obstetric and perinatal outcomes.
- A recent study by Alson et al. (2025) showed lower cumulative success over multiple cycles, especially when adenomyosis is present. Read more in my post: Lower success rates with endometriosis or adenomyosis after 3 IVF cycles.
These findings can be difficult to interpret, partly because of how endometriosis is diagnosed. Studies that use laparoscopy (the gold standard) often include patients who are treated at the same time, which may improve outcomes and mask the true effect of the condition.
Endometriosis is frequently underdiagnosed because it usually requires surgery to confirm. Studies have shown that about 50% of patients with unexplained infertility may have endometriosis and about 25% of patients with RPL may have adenomyosis.
Additionally, some patients have silent endometriosis with no symptoms, making it harder to detect.
Read more in my posts: Nearly half of unexplained infertility patients have endometriosis; Study finds nearly 25% of recurrent pregnancy loss patients have adenomyosis; Explaining unexplained infertility through non-surgical endometriosis diagnosis.
Uterine structural abnormalities
Structural abnormalities of the uterus can interfere with embryo implantation by physically blocking implantation sites or distorting the endometrial cavity.
Common examples include polyps, fibroids (myomas), adhesions (scar tissue), and uterine anomalies such as a septum. These can disrupt the surface where an embryo needs to attach.

Some abnormalities may not be detected with routine imaging. In one study, 44.9% of women with recurrent implantation failure had abnormalities detected on hysteroscopy, most commonly polyps, and outcomes improved after removal (Cenksoy et al. 2013).
Hysteroscopy is often more sensitive than ultrasound and can both diagnose and treat these issues. A meta-analysis by Wang et al. (2024) found that performing hysteroscopy before IVF was associated with higher pregnancy and live birth rates, although the overall quality of evidence is variable. Read more in my post: Meta-analysis analyzes how hysteroscopy impacts IVF success.
Fibroids are a common example. Larger fibroids that distort the uterine cavity clearly reduce implantation, but even smaller fibroids that don’t distort the cavity may still have an effect.
Fibroids can interfere with implantation in several ways, including:
- Disrupting blood flow to the endometrium
- Altering uterine contractions (peristalsis), which may prevent proper embryo positioning
- Triggering inflammation and immune changes in the surrounding tissue
- Affecting gene expression and hormone signaling needed for the endometrium to become receptive
These effects mean that even when the uterine cavity appears normal, fibroids may still impair implantation by changing how the endometrium functions rather than just its shape.
Read more in my posts: Exploring how fibroids can compromise embryo implantation; Meta-analysis examines impact of small fibroids on pregnancy outcomes.
Other structural abnormalities include:
- A hydrosalpinx is a condition where the Fallopian tube is blocked and fills with fluid, and is associated with reduced implantation rates (Johnson et al. 2010).
- A uterine septum is an extra bit of tissue toward the back of the uterus. Venetis et al. (2014) performed a meta-analysis and found that women with uterine septa had higher levels of miscarriage and that their removal (septal resection) reduced miscarriage rates.
- Endometrial fluid (fluid inside the uterine cavity) has been linked to lower live birth rates when it is still present at the time of transfer. If the fluid resolves before transfer, outcomes may be similar to patients without fluid. Read more in my post: Researchers examine how endometrial fluid before transfer affects live birth rates.
- Uterine contractions (peristalsis) that are too frequent before embryo transfer may reduce the chance of pregnancy. A meta-analysis by Vidal et al. (2025) found about a 50% lower chance of clinical pregnancy in patients with high uterine contractions compared to low. Read more in my post: Meta-analysis examines impact of uterine contractions on embryo transfer outcomes.
- Retained tissue in the uterus (such as retained products of conception) can physically block implantation or disrupt the endometrial environment. A 2024 case report (Harpey et al. 2024) details how a woman’s decade-long infertility was resolved after the removal of retained products of conception from a D&C procedure ten years prior. Read more in my post: Patient overcomes decade-long infertility after removal of retained tissue.
- Previous cesarean delivery may be associated with slightly lower implantation and live birth rates, possibly due to scar defects, fluid buildup, inflammation, or changes in uterine function. Read more in my post: IVF success may be lower after a previous cesarean delivery.
Thrombophilia
Thrombophilia is a group of inherited genetic disorders that affect blood clotting (thrombosis) in the blood vessels. Since successful implantation requires an adequate blood supply to the embryo/fetus, those with thrombophilia might have a reduced ability for successful implantation.
Two genes are often attributed to thrombophilia in implantation failure: factor V (which is a protein involved in blood clotting), and MTHFR. Safdarian et al. (2014) found that having both of these mutations were risk factors for RIF.
Other studies (reviewed by Mrozikiewicz et al. (2021)) have confirmed an association, however some studies have not. A 2021 meta-analysis that involved 9 studies by Zeng et al. found no association with MTHFR and implantation failure.
Immunological issues
The immune system plays an important role in implantation. The embryo is genetically different from the mother, so the immune system needs to tolerate it while still functioning normally. If this balance is disrupted, it may contribute to implantation failure or miscarriage.
At the same time, this is one of the most complex and controversial areas in fertility, with inconsistent findings across studies.
Immune balance and inflammation
Implantation requires a controlled immune response. Too much inflammation may be harmful, but too little may also impair implantation.
Ma and Li 2023 suggest that higher systemic inflammation is associated with worse IVF outcomes, including lower live birth rates and higher miscarriage risk. Read more in my post: High systemic inflammation linked to worsened IVF pregnancy outcomes.
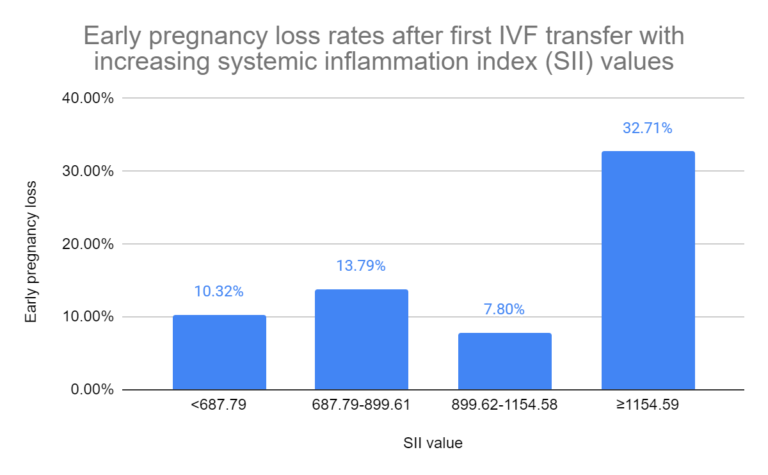
On the other hand, immune activity is also necessary. A very large study by Liu et al. (2024) found that higher levels of certain immune cells before pregnancy were linked to a lower risk of miscarriage, suggesting these cells may support implantation and early pregnancy. Read more in my post: Increases in immune cells associated with reduced risk of miscarriage.
Studies have also found differences in immune-related gene activity in patients with recurrent implantation failure (RIF) or recurrent pregnancy loss (RPL), suggesting that some patients may have an underlying immune-related subtype of infertility. Read more in my posts: Study finds immune signature in endometrium of recurrent pregnancy loss patients; Study shows gene changes in implantation failure, mainly affecting immune cells.
Overall, these results suggest that immune dysregulation may play a role in some patients, but it’s not clear how often this is the true cause or how best to measure it.
Natural killer (NK) cells and HLA compatibility
Natural killer (NK) cells are one of the most studied immune cells in IVF, but also one of the most controversial.
Uterine NK cells (uNK) are abundant at the implantation site and appear to support implantation by helping regulate blood vessel development and placental formation. However, differences in NK cell levels or activity have not consistently been linked to pregnancy outcomes.
There is also interest in how maternal NK cell receptors (KIR) interact with HLA molecules (especially HLA-C) on the embryo. Certain combinations may be less ideal for implantation, although findings are inconsistent and highly individualized.
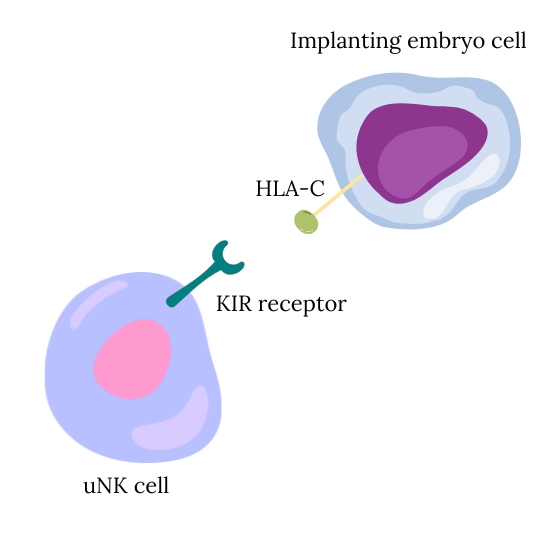
A major issue is that testing methods are not standardized, and there is no clear definition of what constitutes an “abnormal” result. Because of this, interpreting NK cell tests can be difficult and unreliable.
Read more in my post: NK cells in IVF
Autoimmunity
Autoimmune conditions occur when the body produces antibodies against its own tissues. This can create a chronic inflammatory state that could interfere with implantation or early pregnancy.
Some examples include:
- Antinuclear antibodies (ANA): A meta-analysis by Zeng et al. (2019) found that patients who had ANA were more likely to have reduced clinical pregnancy rates, implantation rates, and increased miscarriage rates.
- Antiphospholipid syndrome (APS): an autoimmune disorder that causes an increased risk of blood clots. It is estimated to occur in 5-20% of RPL patients, although this may be overestimated (Vomstein et al. 2021).
While these associations exist, they do not always imply causation, and not all patients with these antibodies experience infertility.
Immunotherapy: mixed evidence
Some IVF treatments aim to modify the immune system, including corticosteroids, heparin, intralipids, IVIG, and others.
There is some evidence that immunotherapy may improve outcomes in a subset of patients with identified immune abnormalities. For example, Khairy et al. (2024) found improved live birth rates in patients with recurrent implantation failure and underlying immunological issues who received treatment. Read more in my post: RIF patients benefit from single or combination immunotherapy during IVF.
However, higher quality evidence tells a different story. A meta-analysis of randomized trials by Melo et al. (2022) found no clear benefit for most commonly used immunotherapies in IVF patients overall. Read more in my post: Meta-analysis of commonly used IVF immunotherapies shows no benefit.
This inconsistency might be due to a key issue in this field: these treatments might only benefit a small subgroup of patients with true, but often undiagnosed, immune-related infertility. Because these patients can be difficult to identify, studies that include the general IVF population tend to show no overall benefit.
There are emerging clinical guidelines in this area. For example, the American Society for Reproductive Immunology (ASRI) recently recommended some immunotherapies, such as corticosteroids, in patients with recurrent pregnancy loss and confirmed immune abnormalities. Read more in my post: ASRI releases 2025 immune therapy guidelines for recurrent pregnancy loss.
However, these recommendations are controversial. The supporting evidence is often limited, and other major organizations (like ASRM and ESHRE) do not currently recommend routine use of immunotherapies due to a lack of high-quality data. Read more in my posts: Italian fertility group issues 2025 guidance on 28 IVF add-ons; Evidence-based recommendations from ESHRE for 27 IVF add-ons.
For a deeper overview of how the immune system works in fertility, see my post: The role of the immune system in infertility and IVF.
Endometrial receptivity
The embryo can only implant into the endometrium when it’s receptive. The optimal time for implantation is the “window of implantation” (WOI).
Endometrial thickness
Endometrial thickness is often used as a marker of receptivity. Thinner linings are sometimes linked to lower live birth rates, but results are inconsistent, and outcomes often plateau once a moderate thickness is reached. Read more in my post: How lining thickness affects success in 30,000 euploid transfers.
Even in large studies, like one by Schmiech et al. (2025), thickness alone is not a strong predictor of success, and pregnancies can still occur with thinner linings. Read more in my post: Study evaluates endometrial thickness and live birth rates in over 244,000 cycles.
Endometrial receptivity array (ERA)
One approach to address this is the endometrial receptivity array (ERA), which analyzes the activity of over 200 genes in the uterine lining to estimate the best time for transfer. This involves a mock cycle followed by a biopsy, and timing is adjusted based on the results.
However, current evidence does not support routine use:
- A meta-analysis by Arian et al. (2022) found no difference in live birth, pregnancy, or miscarriage rates when using the ERA compared to standard timing. Read more in my post: Meta-analysis finds no difference in pregnancy outcomes when using the ERA.
- A randomized trial by Doyle et al. (2022) with euploid transfers also found no improvement in outcomes with ERA-guided timing. Read more in my post: RCT finds no change in birth rates for standard or ERA-timed euploid transfer.
- More recent data by Glujovsky et al. (2026) combined many studies and found no benefit in general IVF patients, although there may be a possible benefit in a subset of patients with recurrent implantation failure using untested embryos. Read more in my post: Meta-analysis finds mixed results for ERA and other receptivity tests.
One limitation is that many studies exclude the patients most likely to benefit (such as those with recurrent implantation failure), making it difficult to draw firm conclusions.
Lifestyle factors
Lifestyle factors can influence both embryo quality and the uterine environment, and may affect implantation, miscarriage, and overall IVF success.
Several factors have been linked to IVF outcomes:
- Smoking has been associated with reduced live birth rates and increased miscarriage risk, with some studies suggesting outcomes similar to women about 10 years older.
- Alcohol use has been linked to fewer eggs retrieved, lower fertilization rates, and higher miscarriage risk.
- Marijuana use has shown mixed results, but some studies suggest an increased risk of pregnancy loss.
- Diet may play a role, with some evidence suggesting benefits from patterns like the Mediterranean diet, although results are inconsistent.
Read more in my post: Lifestyle and egg quality.
Insulin resistance
Insulin resistance (IR) is when the body doesn’t respond properly to insulin, which may affect how the endometrium uses glucose and supports implantation.
It’s commonly linked to PCOS, but can also occur in patients without PCOS or diabetes, often without being detected.
Albert et al. (2025) found that about 30% of IVF patients without PCOS or diabetes had insulin resistance, and these patients had lower live birth rates and higher miscarriage rates, suggesting an effect on the uterine environment.
Testing is relatively simple (for example, with an oral glucose tolerance test), and may help identify patients who could benefit from treatment. In this study, Metformin was associated with improved outcomes in these patients, although more research is needed.
Read more in my post: Silent insulin resistance is common, linked to lower IVF success rates
Clinical and lab factors affecting IVF success
IVF success isn’t only determined by the embryo or uterus. The clinical and lab environment also plays a role, although this is harder to see from the outside.
The embryo transfer
The embryo transfer itself is a critical step. Even with a good embryo, outcomes can vary depending on how smoothly the transfer is performed.
Cirillo et al. (2022) found that ongoing pregnancy rates varied between physicians and embryologists, even after accounting for experience and training.
You can see the ongoing pregnancy rates for each doctor below:

Read more in my post: Embryologist and physician success rates variable, not related to experience.
Tomas et al. (2002) reported on the degree of difficulty in performing a transfer and clinical pregnancy rates. Transfers were difficult if they were time consuming, the catheter met a lot of resistance, there was a need to change the catheter, if cervical dilation was needed, or if blood was found on the catheter. Difficult transfers had a lower clinical pregnancy rate compared to easy transfers (21.1% vs 30.3%).
During embryo transfer, the embryo can become stuck in the catheter and require a retransfer. A meta-analysis by Maleki-Hajiagha et al. (2025) found that retransferring retained embryos was linked to lower pregnancy rates, but note that more research is needed. One study found no differences in pregnancy rates, but showed that retained embryo rate was related to doctor and not to their experience level. Read more in my post: Retained embryos after transfer show comparable pregnancy outcomes.
The IVF lab
The IVF lab environment is tightly controlled because embryos are sensitive to small changes.
Factors like air quality, temperature, oxygen levels, and handling can all affect embryo development. For example, Heitmann et al. (2015) showed that improving air quality can lead to higher implantation and live birth rates.
High-performing clinics tend to follow similar practices, such as culturing embryos under low oxygen, using strict air filtration, and closely monitoring lab conditions. Read more in my post: Common practices of high-performing IVF clinics (2016-2017).
There are also established key performance indicators (KPIs) used internally by clinics, like fertilization rates, blastocyst development, and embryo survival after thawing, to track and maintain quality over time. These metrics aren’t usually visible to patients, but they are routinely monitored within clinics to maintain consistent outcomes. Read more in my post: An Italian consensus on key performance indicators for assessing IVF clinics.
There are also other important factors that can affect outcomes, including:
- Bacterial contamination, which in one case was linked to repeated failed transfers until the issue was resolved. Read more in my post: Contamination linked to repeated failed transfers, patient has live birth after zona removal.
- Equipment or handling issues, such as damage during freezing or thawing. Read more in my post: Frozen embryos damaged after excessive bending of cryostorage device.
- Biopsy of small embryos, with a small study by Singh et al. (2019) showing that this led to lower live birth rates compared to larger embryos. Although more research is needed, this suggests that small embryos may not tolerate the biopsy procedure as well. Read more in my post: Embryo biopsy of small embryos dramatically reduces IVF success rates.
- Exposure to materials in the lab, where plastic consumables may introduce toxicity under certain conditions or alter embryo development, although more research is needed. Read more in my posts: Using multiple plastic consumables in the IVF lab increases risk of embryo or gamete toxicity; Mouse study shows altered placental gene expression when plastic is used to culture embryos.
- Differences between PGT-A testing labs, where Bardos et al. (2022) found that variation in how embryos are analyzed and classified may lead to differences in euploid rates and even live birth rates across labs. Read more in my post: Study finds differences in euploid, live birth rates among PGT-A testing labs.
Conclusion
Implantation and early pregnancy are complex processes, and as you’ve seen, there isn’t a single cause of failure.
In many cases, it comes down to the embryo, especially chromosomal issues. But uterine factors, lifestyle, and clinical or lab conditions can also play a role, particularly when failures happen repeatedly.
Even then, a clear explanation isn’t always found. IVF success isn’t determined by one factor, but by many small pieces working together at the right time. Implantation and early development are complex, multi-step processes that aren’t fully understood. This means that not every failed transfer has a clear explanation, and not every problem can be tested for or fixed.
At the same time, research is continuing to uncover new causes of implantation failure and miscarriage. Wang et al. (2023) discovered 6 genes associated with recurrent miscarriage of euploid embryos, which were found to impair reproductive ability in mouse models. Read more in my post: Whole exome sequencing used to identify genes involved in recurrent euploid miscarriage.
This shows that the field is still evolving, and new insights are continuing to improve our understanding of implantation failure and miscarriage.
If you liked this post and want to support what I do, please consider a paid subscription, Patreon or donate through PayPal!

About Embryoman
Embryoman (Sean Lauber) is a former embryologist and the founder of Remembryo, an IVF research and fertility education website. After working in an IVF lab in the US, he returned to Canada and now focuses on making fertility research more accessible. He holds a Master’s in Immunology and launched Remembryo in 2018 to help patients and professionals make sense of IVF research. Sean shares weekly study updates on Facebook, Instagram, and Reddit regularly. He also answers questions on Reddit or in his private Facebook group.